Ratio detection of inward rectifier potassium channel (Kir) changes in rat basophilic leukemia cells induced by barium chloride
——FLIPR TETRA and voltage sensitive probe
Introduction
FLIPR TETRA and voltage sensitive probes: ratio detection of changes in the inward rectifier potassium channel (Kir) on rat basophilic leukemia cells induced by cesium chloride. Ion channels and transporters are proteins located on the cell membrane that regulate the passage of ions across the biofilm. Studies of these proteins have led to the development of medical treatments for diseases such as arrhythmias, hypertension and neurological disorders. At the same time, these proteins are also potential sites for adverse drug reactions. High-throughput screening methods are highly efficient and cost-effective in producing useful data with minimal amounts of compounds. The FLIPR TETRA® system is equipped with 390–420 nm wavelength excitation LEDs and 440–480 nm and 565–625 nm wavelength filters, using Voltage Sensor Probe (VSP) dyes for high-throughput screening based on the FRET principle. The (HTS) experiment provides a novel cell-based membrane potential change assay. In this experiment, rat basophilic leukemia cells containing an inward rectifier potassium channel (Kir), RBL-2H3, were induced to cause changes in cell membrane potential. Potassium chloride is used to cause depolarization of the cell membrane, and cesium chloride is used to block this depolarization reaction. The ratiometric analysis performed background correction and direct comparison of the FRET data. This application note demonstrates the sensitive and efficient ion channel and transporter-induced changes in cell membrane potential induced by the FLIPR TETRA system in combination with VSP dyes.
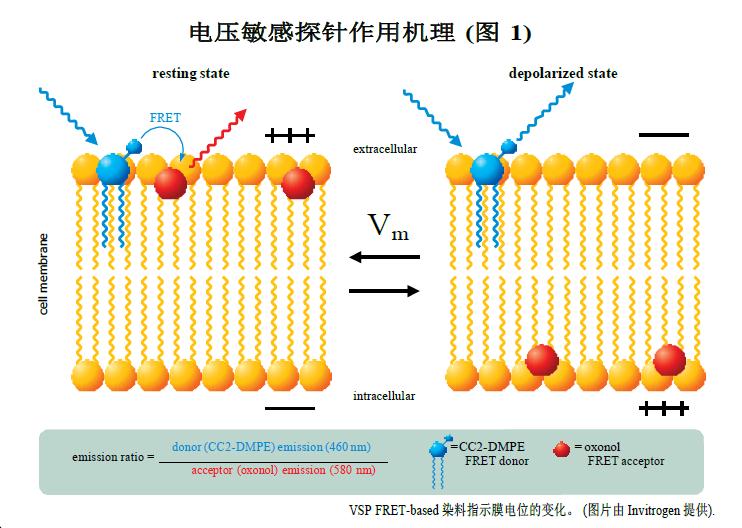
Mechanism of voltage sensitive probe based on FRET
The voltage-sensitive probe is an experimental technique for the development of high-throughput ion channel drugs based on the Fluorescence Resonance Energy Transfer (FRET) principle. The FRET donor, coumarin-phospholipid (CC2-DMPE), binds only to the outside of the cell membrane. The FRET receptor is a mobile, negatively charged hydrophobic cyanine dye compound dye (oxonol) [DiSBAC2 (3) or DiSBAC4 (3)] which binds to one side of the cell membrane depending on the membrane potential change. The resting cell membrane potential is negative. Thus, both probes bind to the outside of the cell membrane to achieve an effective FRET effect. The 390–420 nm wavelength LED excites the CC2-DMPE donor probe and produces a red fluorescent signal at 580 nm from the oxonol receptor probe. When the cell membrane potential is positive, such as KCl-induced cell depolarization, the oxonol receptor probe rapidly changes position to the other side of the membrane. Thus, each oxonol receptor probe "feels" and reacts to changes in cell potential. This transfer position breaks the FRET effect, and the CC2-DMPE donor probe produces a strong blue fluorescent signal at 460 nm after excitation.
material
RBL-2H3: Rat basophilic leukemia cells containing an inward rectifier potassium channel (Kir) (ATCC Cat. #CRL-2256)
Medium: Eagle's Minimum Essential Media (ATCC Cat. #30-2003), 10% FBS (Hyclone Catalog Cat. #SH-30071)
PBS without calcium and magnesium (Invitrogen Cat. #14190-144) Trypsin/EDTA (0.05% Trypsin/1mM EDTA) (Invitrogen Cat. #25300-054)
VSP solution-1: 160 mM NaCl (Sigma Cat. #S-5886), 4.5 mM KCl, (Sigma Cat. #P5405), 2 mM CaCl2 (Sigma Cat. #C-7902), 1 mM MgCl2 (Sigma Cat. #M-4880), 10 mM glucose (Sigma Cat. #G-7021), 10 mM HEPES (Invitrogen Cat. #15630-080), pH 7.4
High potassium solution: 164 mM KCl, 2 mM CaCl 2 , 1 mM MgCl 2 , 10 mM glucose, 10 mM HEPES, pH 7.4
Sterile water (Irvine Scientific Cat. #9309)
DMSO (Dimethyl Sulfoxide, Aldrich Cat. #47,230-1)
Barium chloride (Sigma Cat. #B-0750)
Pluronic F127 (Invitrogen Cat. #P3000)
Voltage Sensitive Probe Kits: DisBac2(3) and CC2-DMPE (Invitrogen Cat. #K1016), VABSC-1 Background Inhibiting Dyes (Invitrogen Cat. #K1019)
FLIPR TETRA High-throughput Fluorescence Detection and Analysis System (Molecular Devices)
method
Cell preparation
Pre-cultured RBL-2H3 cells were trypsinized and washed and seeded in BD BioCoatTM polylysine coated 384-well plates (Cat. #356663, BD Biosciences) 18,500 before each experiment. Cells. The cells were cultured overnight at 37 ° C and 5% CO 2 . The mother liquor, dye and buffer can be placed at room temperature on the day of the experiment. VSP loading buffer and cesium chloride concentration gradient compounds were prepared in VSP-1 buffer.
Prepare VSP loading buffer
CC2-DMPE buffer:
Step 1 Prepare 5 mM DMSO stock solution and store in aliquots at -20 °C.
Step 2 On the day of the experiment, prepare a working solution. The CC2-DMPE mother liquor was mixed with the same volume of 10% Pluronic Acid and subsequently diluted to 5 μM with VSP-1 solution.
Step 3 The buffer is thoroughly mixed and protected from light before use.
DiSBAC2 (3) buffer:
Step 1 Prepare 12 mM DiSBAC 2 (3) DMSO stock solution and store to -20 ° C.
Step 2 Prepare the water mother liquor of 200 mM VABSC-1 and store it in the dark at room temperature. For more instructions, refer to the Invitrogen VSP Product Brochure.
Step 3 On the day of the experiment, a working solution of 4 μM DiSBAC 2 (3) containing 0.2 μM VABSC-1 quencher was prepared using VSP-1 solution. VSP dye experiments in the FLIPRTETRA system have no special requirements other than the installation of corresponding LEDs and filters.
Loading dye in cells
Step 1 Remove the medium from all wells in the 384-well plate and add 50 μL of VSP-1 solution instead.
Step 2 Immediately remove the VSP-1 solution and add 25 μL of CC2-DMPE loading buffer.
Step 3 Incubate the cell plate at room temperature for 30 minutes in the dark.
Step 4 After 30 minutes, remove CC2-DMPE loading buffer and add 50 μL of VSP-1 solution.
Step 5 Immediately remove VSP-1 and add 25 μL of 4 μM DiSBAC 2 (3) and 0.21 μM VABSC-1 loading buffer.
Step 6 Add 10X concentration of BaCl2 compound (ion channel inhibitor) to the corresponding well
Step 7 Incubate the cell plate for 30 minutes at room temperature in the dark. Refer to the Invitrogen VSP User Guide for more details.
FLIPR TETRA system configuration and calibration
The FLIPRTETRA system's operating software, ScreenWorks., has a very user-friendly interface for easy editing and parameter setting of experimental protocols. More information can be found in the FLIPRTETRA User Guide. The system comes with a yellow detection plate that can be used to calibrate 390–420 nm excitation source LEDs and 565–625 nm emission filters. A test plate containing 7-diethylamine coumarin-3-carboxylic acid was used to calibrate the 390-420 nm excitation source LED and the 440-480 nm emission filter, as described below.
390–420 nm excitation source LED and 440–480 nm filter calibration plate preparation
Step 1 A 0.02 M solution of 7-diethylamine coumarin-3-carboxylic acid DMSO (Molecular Probes Cat. #D-1421) was prepared.
Place in a centrifuge tube and vortex to mix.
Step 2 is divided into small portions and stored at -20 °C.
Step 3 Dissolve a second step of the mother liquor within 1 hour of calibration and dilute to 20–30 mL 10-5 M solution in the same buffer for dye loading. Adjust the pH to 7.4 and vortex to mix.
Step 4 Add 25 μL (384-well plate) or 100 μL (96-well plate) of the coumarin solution to each well.
Running VSP experiments in the FLIPRTETRA system
Parameters such as the volume of the experimental solution, the height of the sample, and the speed are optimized depending on the specific experiment and cell type requirements to avoid blowing the cells at the bottom of the well.
Table 1 is a recommended RBL-2H3 cell experimental machine parameter setting for reference.
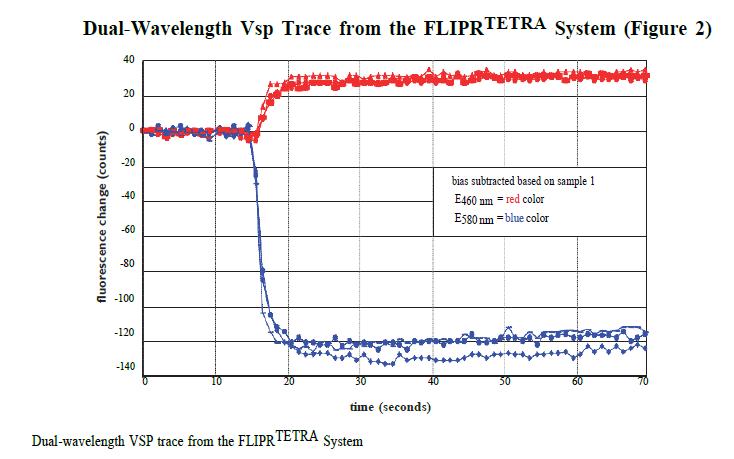
In the FLIPR TETRA system, signals of 440–480 nm and 565–625 nm before the addition of 25 μL of high K+ depolarization solution were recorded for 10 seconds and recorded again for 10 seconds 40 seconds after loading. The average signal records at these two time points in the experiment form the basis of the ratio analysis.
Ratio data calculation
Ratio data analysis provides a direct comparison of background correction and FRET's 440–480 nm and 565–625 nm data. Data results for the same cell can be compared in both polarized and non-polarized states. Figure 2 shows an example of a set of data from the FLIPR TETRA system.
In the same data window, the average signal value of 40-60 seconds before and after loading was detected. The average signal values ​​in the cell-free recording wells were removed as baseline corrected values ​​(BLC) at the time of the statistics. The standard ratio value (R / Ro) of the reaction background calibration was calculated by dividing the RFU value after adding the depolarization effect agent by the initial RFU value in each well.
Ratio of emitted light in depolarized and polarized states:
Emission RatioPolarized = BLC 440–480initial/BLC 565–625initial
Emission RatioDepolarized = BLC 440–480final/BLC 565–625final
The response ratio is calculated as:
R/Ro= Emission RatioDepolarized/Emission RatioPolarized
result
Using the FLIPRTETRA system, it was found that the IC50 of guanidine chloride inhibiting Kir on rat basophilic leukemia cells was 270 μM. This result was calculated using a 4-parameter curve on Graphpad Prism® software. The data obtained by the conventional patch clamp results in an IC50 of 500 μM. As an evaluation of the overall performance of the experiments performed on the FLIPRTETRA system, the Z factor calculated at the IC80 level was 0.58. The Z factor is a tool for comparing and evaluating the quality of an experiment and for optimizing the experiment. 3 Z factor > 0.5 is considered to be an effective experiment.
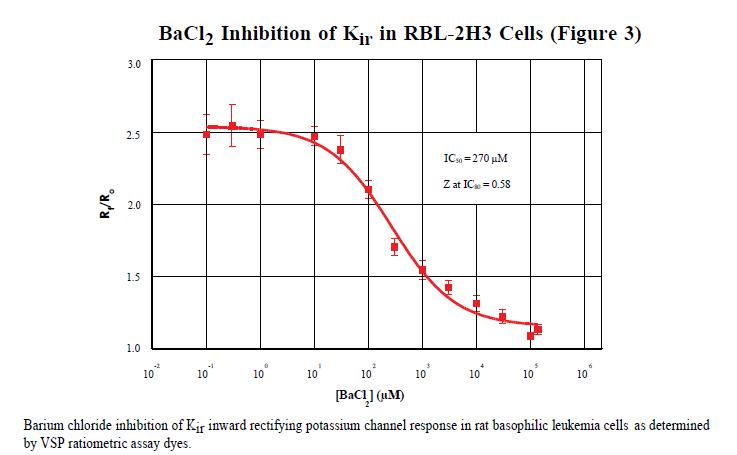
in conclusion
The FLIPRTETRA system is equipped with 390–420nm excitation source LEDs and 440–480nm and 565–625nm filters for efficient voltage-sensitive probe experiments with ratiometric detection. The IC50 results are consistent with the results of traditional patch clamp techniques. The ratiometric dye experiment and the FLIPRTETRA system, along with other membrane potential reagents, are important tools for ion channel functional assays and experimental studies. A variety of user-replaceable LED modules and filters combined with ratiometric dyes and real-time recording of 96-well and 384-well recording modes provide very flexible ion channel and transporter-induced changes in membrane potential.
references:
1. M. Falconer, et al. High-throughput screening for ion channel modulators.
J Biomol Screen Volume 7, (5) 460–465 (2002).
2. J. Gonzalez, et al. Cell-based assays and instrumentation for screening ion-channel targets. Drug Discovery Today 4(9): 431–439 (1999).
3. J. Zhang, et al. A simple statistical parameter for use in evaluation and validation of high-throughput screening assays. J Biomol Screen Vol. 4, (2) 60–73 (1999).
Anesthesia Medical Co., Ltd. , https://www.medicaldiverse.com