This application note shows how to grow adherent cells in cell culture microchannels. Cell seeding, medium exchange, and optical properties will be described. In addition, the main differences between cell culture channels and standard open well formats are shown.

2. Medium exchange
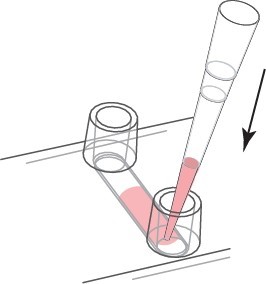
a. Continuous Media Exchange - Recommended For continuous media replacement, first remove the old media from the storage. Then, an appropriate amount of fresh medium is added to one reservoir while aspirating from the other reservoir. Carefully use the cell culture pipette. We recommend a volume of at least 3 times the channel volume. Refill the reservoir.
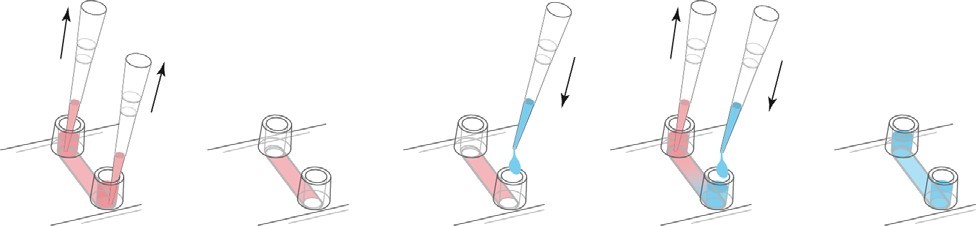
The medium of continuous exchange medium is 3 times the volume.
b. Complete culture fluid exchange - only for expensive liquids
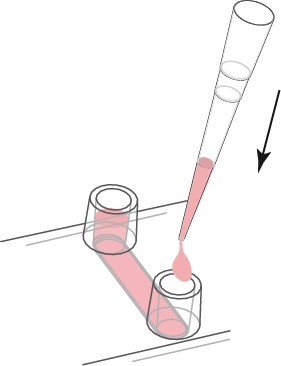
If you only want to change the liquid in the channel, first empty the channel. The tip of the pipette is then placed on the channel inlet and the liquid is carefully aspirated from the channel. Use a cell culture pipette to thoroughly remove all liquids. In order to refill the channel, fresh material of the channel capacity is injected directly into the channel. Avoid creating bubbles. Refill the reservoir holes on both sides.
Clearing the channel completely may result in bubbles forming after refilling.

The channel and reservoir holes are completely replaced.
3. Channel Slides and Porous Slides <br> In the following section, we compare the characteristics of μ-Slide VI 0.4 (cell culture channels) with μ-Slide 8 wells (open format).
For cell seeding, different cell densities must be applied to obtain the same amount of cells on the surface. In this example, the goal is to inoculate 25 cells per unit area. Since the height of the liquid in the μ-Slide 8 well is 7.5 times, the applied cell concentration must be lower than the same factor.
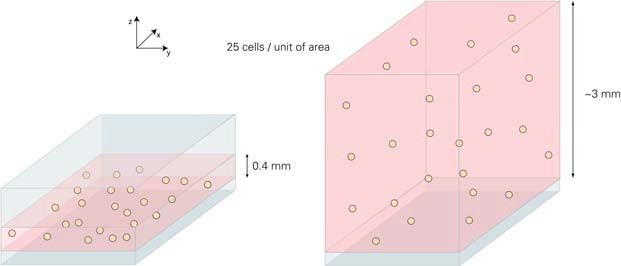
Liquid internal μ-Slide VI 0.4 and μ-Slide 8 holes based on different heights. In the above example, the same growth area and number of cells but different volumes result in different cell concentrations.
In order to achieve a considerable degree of fusion, we recommend using 3 ... 7 x 105 cells / ml (μ-Slide VI 0.4) and 4 ... 9 x 104 cells / ml (μ-Slide 8 wells). This is the same ~7.5 times between the two geometries. After cell adhesion, the number of cells per unit area is the same.
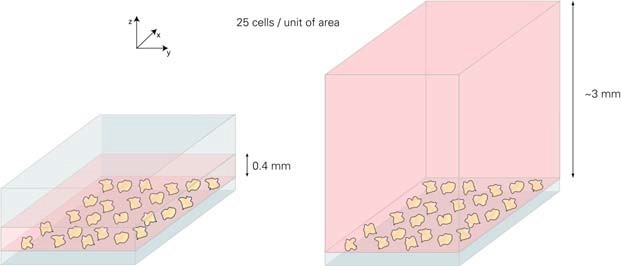
Due to the different heights, different cell concentrations produce the same level of cellular aggregation.
4. Advantages of Channel μ-Slide <br> Channel slides have a strong advantage over standard aperture formats.
a. Phase contrast microscopy is better. The channel geometry is very convenient to use phase contrast microscopy. Because there is no influence of the concave surface, the whole growth area can be observed by phase contrast technique.

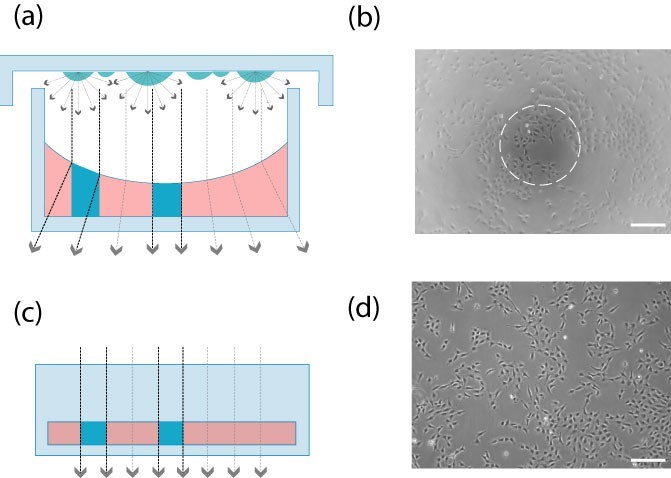
Open perforated plates (Fig. a, Fig. b): The concave surface affects the phase difference effect. Only the center provides high quality contrast.
Channel slides (Figure c, Figure d): In the channel geometry, the beam paths are always aligned. High quality phase contrast microscopy is available throughout the field of view.
b. Evenly distributed cells
In the channel, the homogeneity of the adherent cells is much better because of the small edge effect.
In the microscopic images below, the phase difference (a) and fluorescence (b) modes show the heterogeneity of cell distribution in the open wells (1-3) and the homogeneity in the cell culture channels (4-6).

2 open porous slides, random position imaging
3 open porous slides, central part
4-channel slide, edge position imaging
5 channel slides, random position imaging
6-channel slide, center position imaging
Anti-insect And Anti-bird net,Nets To Protect Plants,Agricultural Anti Bird Mesh Netting
Changzhou Green Nets Co.,Ltd. , https://www.czglnets.com