The longest follow-up of over 1,000 people, the second-line treatment of lung cancer is overwhelming!
May 10, 2019 Source: Singularity Network
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Four years ago, data on the treatment of melanoma with the immunological checkpoint inhibitor ipilimumab targeting CTLA-4, for the first time, the world has seen the "excessive standby" ability of immunotherapy [1].
Two years ago, we saw the power of immunotherapy in patients with advanced non-small cell lung cancer (NSCLC).
At the 2017 American Association for Cancer Research (AACR) annual meeting, the 5-year follow-up data from the CheckMate-003 study was amazed: overall, the 5-year survival rate for patients with advanced NSCLC who received PD-1 antibody nivolumab was 16% [ 2]. The "5-year survival rate of 16%" that is often mentioned in the field of tumor immunity is derived from this.
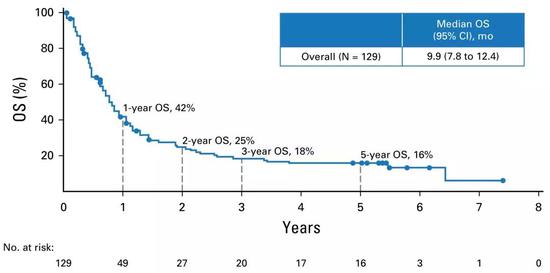
â–² 5-year survival curve of CheckMate-003 [2]
Come and watch the "5-year survival rate of 16%" from the above picture
What does this data mean? We have to look at the 2008-2015 data from the National Cancer Institute: the 5-year survival rate for patients with advanced NSCLC is only about 5% [3].
The five-year survival rate of patients with advanced NSCLC who received nivolumab treatment was more than three times that of historical data.
Not only that, but the researchers also found in two Phase III clinical trials, CheckMate-017/057, that patients receiving nivolumab had a longer-term benefit than patients receiving docetaxel chemotherapy [4].
Acquiring the above research results not only did not stop the researchers, but also stimulated their curiosity and enthusiasm: can the above research results be reproduced after a longer follow-up period in a larger patient population?
The answer was finally revealed last month!
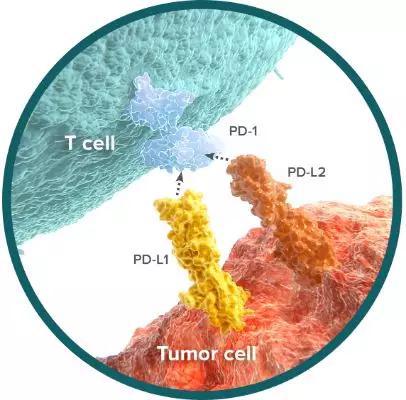
â–² PD-1/PD-L1 path
It was at last month's AACR annual meeting that 17 research institutes jointly released the largest and most long-term follow-up PD-1 antibody treatment study [5].
The investigators summarized long-term follow-up data from four clinical trials, CheckMate-003/063/017/057. Of the total 1,100 patients, 54% received first-line treatment before receiving nivolumab, and even 9% received 4 or more treatments. In the pooled analysis of CheckMate017/057, 80% and 82% of patients receiving nivolumab and docetaxel had received first-line treatment.
Even in such a large population, patients who received nivolumab treatment maintained a 14-year overall survival rate of 14%. In addition, you can also see in the figure below that from the third year on, the patient's survival will enter the platform period, and the benefits will continue to exist.
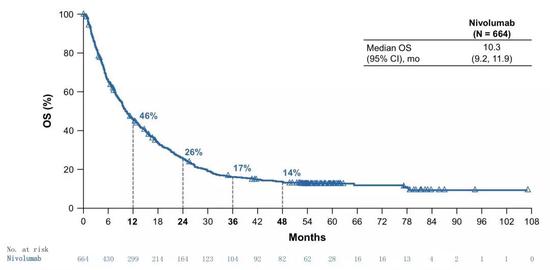
â–² megacurve super curve [5]
In this ultra-large-scale, long-term follow-up analysis, nivolumab's "combat angels" feature was unfolded: landmark analysis showed complete remission (CR) and partial remission (PR) within 6 months of docetaxel treatment The 4-year overall survival rate was only 12% for patients, and the amazing 5% for the nivolumab group, which is nearly five times that of the docetaxel treatment group. (Seeing this data is not very curious what is the landmark analysis? I will tell you later.)
Nivolumab's performance is still strong and has withstood a large sample size test.
â–² Pathological section of non-small cell lung cancer (speakinghealth.com)
This summary analysis included the previous four nivolumab important studies. Phase 1 study CheckMate-003 began as early as 2008, and the optimal clinical dose of nivolumab was determined to be 3 mg/kg in multiple solid tumors [2,6]. The next phase 2 clinical study, CheckMate-063, was revalidated [7].
CheckMate-017 [8] and CheckMate-057 [9] were phase 3 clinical studies conducted in the same period in 2012, targeting squamous NSCLC and non-squamous NSCLC patients, and patients in the chemotherapy group receiving docetaxel. Both studies confirmed that patients who received nivolumab had a significantly better overall survival than docetaxel chemotherapy.
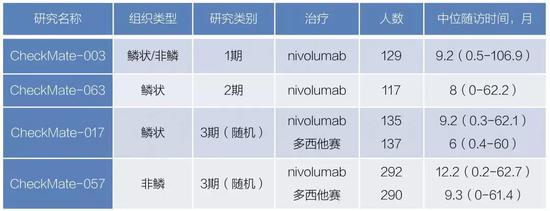
â–² Introduction to four clinical studies [5]
The milestone clinical research results above have been published for some time. Are those patients still okay? This is what this megacurve data analysis of the oversized patient population wants to know.
As mentioned above, in the four clinical studies with a cumulative number of over 1,000 people and a short follow-up of 51.6 months, the 4-year overall survival rate of patients receiving nivolumab treatment still reached 14%, which is not easy.
Since the chemotherapy control was set in the two phase 3 clinical trials of CheckMate-017/057, to compare the difference between the efficacy of nivolumab and docetaxel chemotherapy, long-term follow-up data from the two phase 3 clinical studies were analyzed.
The results were as follows: the 4-year overall survival rate of the docetaxel chemotherapy group was only 5%, which was as low as ever; and the nivolumab-treated patients had a 4-year overall survival rate of 14%, which was consistent with the overall analysis. .
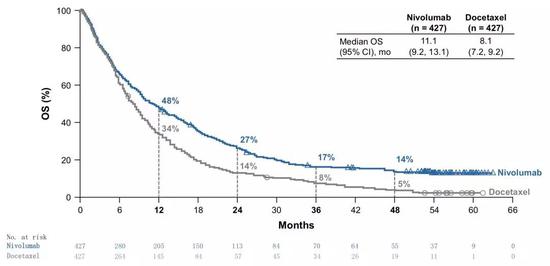
â–² CheckMate-017/057 data [5]
The next question the researchers thought was: What is the relationship between the different types of responses and the long-term survival benefits of patients in the nivolumab and docetaxel-treated groups?
In order to answer this question more scientifically, this analysis introduced the landmark landmark analysis method [10]. This method was widely used as early as the 1980s. It was originally discovered in the field of transplantation that patients in the experimental group need to wait for the waiting period of treatment, and there can be no events during the period. Such patients may have "advantages" themselves.
Similarly, the same was true for patients who eventually “responded†during long-term follow-up. The existence of this "time-to-response" bias can be erroneously beneficial to alleviate the living conditions of the population.
The definition of a "landmark" stipulates that patients who died before this time point are not included in the follow-up analysis, and patients who respond after the landmark time point are not included in the subsequent analysis to effectively reduce this bias [11]. In this way, the test group and the control group can compete for treatment under fair conditions.
This is also the original intention of introducing landmark analysis in this study.
â–² Source: pharmatimes.com
By analyzing CheckMate-017/057 data, the researchers found that most of the patients' response to nivolumab treatment and docetaxel chemotherapy appeared during the first 6 months of treatment. Therefore, it is reasonable to use the sixth month after treatment as the landmark point.
After the introduction of the landmark point, the researchers divided the two groups into four types: complete response (CR), partial response (PR), stable disease (SD), and disease progression (PD) based on the patient's response to treatment.
Researchers who see the data should be as shocked as the singularity.
For those with CR and PR, the 4-year survival rate is as high as 58%, which means that as long as patients with advanced NSCLC respond to nivolumab treatment during the first 6 months of treatment, most patients can survive for more than 4 years. It is nearly 5 times more than the treatment of docetaxel chemotherapy!
Moreover, for this group of patients, the median sustained response time of docetaxel chemotherapy is only half a year, and the nivolumab treatment lasts for 2 years, and the "excessive standby" ability is once again demonstrated.
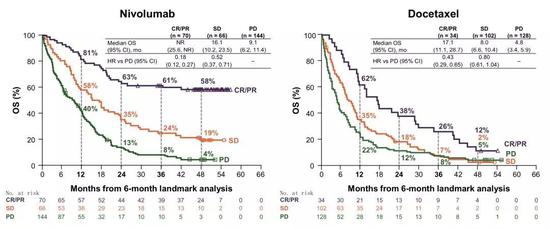
â–² landmark analysis CheckMate-017/057 data [5]
Even those who did not respond to nivolumab treatment but remained stable, their 4-year survival rate was as high as 19%, and this data turned out to be nearly 10 times that of docetaxel chemotherapy!
This data is somewhat surprising because we are accustomed to believe that they cannot benefit from immunotherapy for patients whose tumor volume remains constant after treatment and their condition remains stable. This set of data shows that SD patients can not only benefit, but also benefit more than imagined.
It now appears that as long as the condition does not progress within 6 months of treatment, there is hope for benefit from immunotherapy - no progress, that is, benefit. In fact, in the CheckMate-003 study we introduced separately, two of the 16 patients who were followed up for 5 years after nivolumab treatment were SD patients, and their condition remained until the last study database was locked. Relieve the status.
â–² Source: Anne Weston, LRI, CRUK. Wellcome Images. Boston University
Another phenomenon that deserves attention is that patients with CR/PR and SD who are treated with nivolumab, even if their condition progresses, the 3-year survival rates of those who progress are still 26% and 13%, respectively, are docetaxel chemotherapy. Corresponding to 2 times the grouping. It also shows that patients who benefit from immunotherapy, even if the disease progresses, will be slower than chemotherapy.
Data from these different types of response indicate that we can no longer measure the efficacy of immunotherapy with the objective response rate in the era of chemotherapy or targeted therapy. After all, from the mechanism of action, immunotherapy is very different from chemotherapy and targeted therapy.
It is understood that this is the first time in such a large population of patients with advanced NSCLC that the benefits of nivolumab to patients are more durable than docetaxel chemotherapy - once effective, long-lasting benefits - even if the patient's condition is here Then there will be progress.
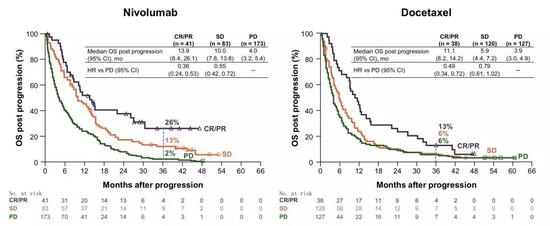
â–² Patients receiving SR/PR and SD treated with nivolumab benefit even if the disease progresses eventually [5]
As for the long-term safety of nivolumab treatment, the researchers also analyzed the data from these four clinical studies. Overall, patients were well tolerated with nivolumab, and no new adverse events were found in subsequent follow-up data. Moreover, adverse events mainly occurred in the first year, and then decreased year by year, and the most important adverse reaction was fatigue.
"These results were generated after analysis of a large number of patients with advanced non-small cell lung cancer, and revealed for the first time that the patient's response to nivolumab was associated with years of survival benefit." After seeing the data from this study, Duke cancer Scott Antonia, director of the Center for Cancer Immunotherapy, commented [12], "Because these patients had a five-year survival rate of only 5%, the results of this long-term survival summary analysis made more sense!"
Singularity cake wants to say that since Nivolumab was approved by the US FDA for the second-line treatment of metastatic NSCLC in 2015, it has continued to surprise us. In June last year, nivolumab was also approved by the China National Drug Administration to open the era of immunotherapy in China at the lowest price in the world.
Even when nivolumab was not listed in China last year, it was included in the 2018 version of CSCO's primary lung cancer diagnosis and treatment guidelines because of its sufficient evidence, as a Class III recommendation for non-driven gene NSCLC second-line treatment (Class 1 evidence). And just in the 2019 CSCO guide just released last week, nivolumab's recommendation level was raised to a level I recommendation with a level of evidence of 1A .
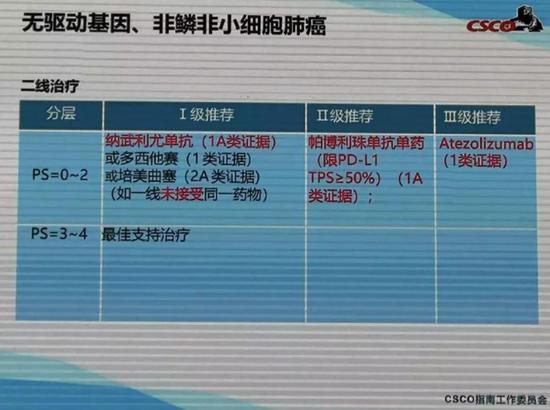
â–² 2019 CSCO Guide Releases Live Slides
All in all, this large sample, long-term follow-up of the “megacurve super curve†study shows that patients benefit more from nivolumab treatment than docetaxel chemotherapy.
More importantly, the study also found that as long as the patient's condition remains stable after treatment, it is expected to benefit for a long time, which to a certain extent subverts the traditional tumor efficacy evaluation method.
In any case, nivolumab, as the standard treatment for post-chemotherapy in patients with advanced NSCLC, will hope for more patients to survive in the future.
Reference materials:
[1].Schadendorf D, Hodi FS, Robert C, et al. Pooled Analysis of Long-Term Survival Data From Phase II and Phase III Trials of Ipilimumab in Unresectable or Metastatic Melanoma [J]. Journal of Clinical Oncology, 2015, 33 (17): 1889-1894. DOI: 10.1200/JCO.2014.56.2736
[2] Gettinger SN, Horn L, Jackman DM, et al. Five-Year Follow-Up of Nivolumab in Previously Treated Advanced Non–Small-Cell Lung Cancer: Results From the CA209-003 Study[J]. Journal of Clinical Oncology , 2018, 36(17): 1675-1684. DOI: 10.1200/JCO.2017.77.0412
[3]. Noone AM, Howlader N, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2015, National Cancer Institute. Bethesda, MD [J]. 2018.
[4].Horn L, Spigel DR, Vokes EE, et al. Nivolumab versus docetaxel in previously treated patients with advanced non–small-cell lung cancer: Two-year outcomes from two randomized, open-label, phase III trials (CheckMate 017 and CheckMate 057) [J]. Journal of Clinical Oncology, 2017, 35(35): 3924. DOI:10.1200/JCO.2017.74.3062
[5]. Julie Brahmer, Hossein Borghaei, et al. Long-term survival outcomes with Nivolumab in previously advanced advanced non-small cell lung cancer patients: impact of early disease control and response [C].
[6].Gettinger SN, Horn L, Gandhi L, et al. Overall survival and long-term safety of nivolumab (anti-programmed death 1 antibody, BMS-936558, ONO-4538) in patients with previously advanced advanced non–small -cell lung cancer[J]. Journal of clinical oncology, 2015, 33(18): 2004. DOI:10.1200/JCO.2014.58.3708
[7].Rizvi NA, Mazières J, Planchard D, et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063) : a phase 2, single-arm trial[J]. The Lancet Oncology, 2015, 16(3): 257-265. DOI:10.1016/S1470-2045(15)70054-9
[8]. Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer[J]. New England Journal of Medicine, 2015, 373(2): 123- 135. DOI: 10.1056/NEJMoa1504627
[9].Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer[J]. New England Journal of Medicine, 2015, 373(17): 1627- 1639. DOI: 10.1056/NEJMoa1507643
[10].Dafni U. Landmark Analysis at the 25-Year Landmark Point[J]. Circulation-cardiovascular Quality and Outcomes, 2011, 4(3): 363-371. DOI:10.1161/CIRCOUTCOMES.110.957951
[11]. Anderson JR, Cain KC, Gelber RD, et al. Analysis of survival by tumor response. [J]. Journal of Clinical Oncology, 1983, 1(11): 710-719. DOI: 10.1200/JCO.1983.1 .11.710
[12]https://news.bms.com/press-release/corporatefinancial-news/bristol-myers-squibb-announces-long-term-survival-results-pool
NINGBO MEDICAL EQUIPMENT CO.,LTD , https://www.techartmeds.com