Human prostaglandin E2 ( PGE2 ) ELISA kit
Product No:EH4233 Size: 48T/96T
Specificity: human range: 31.2-2000pg/ml
Sensitivity: <18.75pg/ml
Application: For the quantitative detection of PGE2 in serum, plasma, tissue homogenate and other biological fluids.
Storage: 4 ° C, 6 months Note: For research use only.
Kit component
| Item | Specifications(48T/96T) | Storage |
| MicroELISAPlate(Dismountable) | 8×6or8×12 | 4°C/-20°C |
| LyophilizedStandard | 1vialor2vial | 4°C/-20°C |
| Sample/Standarddilutionbuffer | 10ml/20ml | 4 ° C |
| Biotin-detectionantibody(Concentrated) | 30ul/60ul | 4 ° C |
| Antibodydilutionbuffer | 5ml/10ml | 4 ° C |
| HRP-StreptavidinConjugate (SABC) | 60ul/120ul | 4°C (shadinglight) |
| SABCdilutionbuffer | 5ml/10ml | 4 ° C |
| TMBsubstrate | 5ml/10ml | 4°C (shadinglight) |
| Stopsolution | 5ml/10ml | 4 ° C |
| Washbuffer(25X) | 15ml/30ml | 4 ° C |
| PlateSealer | 3/5pieces | |
| ProductDescription | 1copy |
Principle of Analysis <br> This ELISA kit uses a competitive ELISA as a method. The microtiter plates provided in the kit have been pre-coated with PGE2. During the course of the reaction, PGE2 competes with a fixed amount of PGE2 for biotinylation of PGE2-specific antibodies on a solid support in a sample or standard. Excess conjugated and unbound samples were either washed away from the plates and HRP-streptavidin (SABC) was added to each well to incubate. The TMB substrate solution was then added to each well. The enzyme substrate was added to the sulfuric acid solution to terminate the reaction, and the color change was measured spectrophotometrically at a wavelength of 450 nm. The concentration of PGE2 was then determined by comparing the OD of the sample to a standard curve.
Precautions
1. In order to verify the validity of the experimental operation and the appropriateness of the sample dilution ratio, it is recommended to use a standard product and a small amount of sample for the experimental test.
2. Keep the board dry after opening and before use.
3. Before using the kit, rotate the tube and lower all components down the bottom of the tube.
4. Store TMB reagents protected from light.
5. The washing process is very important, and it is easy to cause false alarms if it is not fully washed.
6. For standard and sample testing, duplicate well assays are recommended.
7. Do not allow the Microplate to dry during the measurement as the dry plate deactivates the active components on the plate.
8. Do not reuse tips and tubes to avoid cross-contamination.
9. Avoid using different batches of reagents together.
Required material but not provided
1. Microplate reader (wavelength: 450nm)
2.37 ° C incubator
3. Automatic washing machine
4. Precision single and multi-channel pipettes and disposable tips
5. Cleaning tube and Eppendorf tube
6. Deionized or distilled water
Manual cleaning <br> Discard the solution in the plate without touching the side walls. Take the board on an absorbent filter or other absorbent material. Each well was completely filled with 350 ul of wash buffer and soaked for 1 to 2 minutes, then the contents were aspirated from the plate and the plate was taken on an absorbent filter paper or other absorbent material. This process was repeated twice and washed a total of three times.
Automated Cleaning <br> Aspirate all wells and wash the plate three times with 350 ul of wash buffer. After the last wash, turn the plate and pat the plate on absorbent filter paper or other absorbent material. It is recommended to set the gasket to soak for 1 minute.
Sample Collection and Storage <br> Test samples are separated immediately after collection and immediately analyzed (within 2 hours). Or aliquot and store at -20 °C for a long time. Avoid multiple freeze-thaw cycles.
Serum: The whole blood sample was allowed to stand at room temperature for 2 hours or placed at 4 ° C overnight, centrifuged at about 1000 x g for 20 minutes, and the supernatant was collected and assayed immediately. The blood collection tube should be disposable, pyrogen-free and non-endotoxin.
Plasma: Plasma was collected using EDTA-Na2 as an anticoagulant. The sample was centrifuged at 1000 x g for 15 minutes at 2-8 ° C within 30 minutes after collection. The supernatant was collected and assayed immediately. Avoid hemolysis, high cholesterol samples.
Tissue homogenization: Since hemolyzed blood is associated with the test results, residual blood must be removed by washing the tissue with pre-cooled PBS buffer (0.01 M, pH = 7.4). After weighing, the tissue was minced and homogenized in PBS (volume depends on the weight of the tissue. In general, 9 mL of PBS is suitable for 1 gram of tissue block. Some protease inhibitors are recommended for addition to PBS) with glass on ice. Chemist. To further destroy the cells, you can sonicate the suspension with an ultrasonic cell disruptor or place it in a freeze-thaw cycle. The homogenate was then centrifuged at 5000 x g for 5 minutes to obtain a supernatant.
Cell culture supernatant: The supernatant was centrifuged at 1000 x g for 20 minutes at 2-8 ° C to remove insoluble impurities and cell debris. The clear supernatant was collected and tested immediately.
Other biological fluids: Centrifuge at 1000 xg for 20 minutes at 2-8 °C. The supernatant was collected and assayed immediately.
Sample preparation: The sample should be clear and transparent and centrifuged to remove suspended solids.
Note: Samples used within 5 days can be stored at 4 ° C. In addition, samples must be stored at -20 ° C (analysis ≤ 1 month) or -80 ° C (analysis ≤ 2 months) to avoid loss of biological activity and contamination. Hemolyzed samples are not suitable for this assay.
Sample Dilution Guidelines <br> The end user should estimate the concentration of the target protein in the test sample and select the appropriate dilution factor so that the diluted target protein concentration falls within the optimal detection range of the kit. Dilution of the sample with the supplied dilution buffer may require multiple tests. The test sample must be thoroughly mixed with the dilution buffer. Standard curves and samples should also be prepared in the pre-test. The following dilution solutions are for reference only:
High concentration ( 20000-200,000 pg/ml ): Dilution: 1:100 (ie, 1 μl of sample is added to 99 μl of sample/standard dilution buffer.)
Medium concentration ( 2000-20000 pg/ml ): Dilution : 1:10 (ie, 10 μl of sample is added to 90 μl of sample/standard dilution buffer.)
Low concentration ( 31.2-2000pg/ml ): Dilution: 1:2 (ie 50 μl sample is added to 50 μl sample/standard dilution buffer).
Very low concentration ( ≤31.2pg/ml ): no dilution or 1:2 dilution.
Reagent Preparation and Storage <br> Place the kit at room temperature for 20 minutes before use.
1 , washing buffer:
50 mL of concentrated wash buffer was diluted to 750 mL of wash buffer with deionized or distilled water. The unused solution was returned to 4 °C. If crystals are formed in the concentrate, they can be heated in a 40 ° C water bath (heating temperature should not exceed 50 ° C) and gently mixed until the crystals are completely dissolved. The solution should be cooled to room temperature before use.
2 , standard:
1). 100 ug/ml standard solution: 1 ml sample/standard dilution buffer was added to a standard tube, the tube was kept at room temperature for 10 minutes and thoroughly mixed.
2). 50 ug / ml → 1.563 ug / ml standard solution: labeled 6 Eppendorf tubes, respectively, 1000pg / ml, respectively 500pg / ml, 250pg / ml, 125pg / ml, 62.5pg / ml, 31.2pg / ml. Add 0.3 ml sample/standard dilution buffer to each tube. 0.3 ml of the above 100 ug/ml standard solution was added to the first tube and mixed well. Transfer 0.3 ml from the first tube to the second tube and mix thoroughly. Transfer 0.3 ml from the 2nd tube to the 3rd tube and mix thoroughly, and so on.
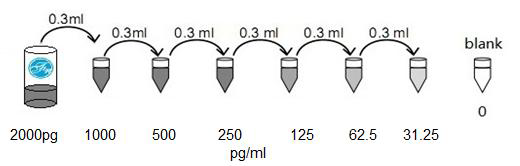
Note: It is best to use a standard solution within 2 hours. The standard solution should be between 4 ° C and 12 hours. Or store it at -20 ° C to 48 hours. Avoid repeated freeze-thaw cycles.
3. Preparation of biotin-labeled antibody working solution <br> Prepared within 1 hour before the experiment.
1). Calculate the total volume required for the working solution: 0.05 ml / hole × number of wells.
(0.1-0.2 ml more than the total volume)
2). Dilute the biotin detection antibody 1:10 with antibody dilution buffer and mix thoroughly.
(Import 1 μl of biotinylated antibody into 99 μl of antibody dilution buffer.)
4, HRP- streptavidin, avidin preparation (the SABC) working solution in combination:
Prepare within 30 minutes of the experiment.
1). Calculate the total volume required for the working solution: 0.1 ml / hole × number of holes.
(0.1-0.2 ml more than the total volume)
2). Dilute SABC with 1:100 SABC Dilution Buffer and mix thoroughly.
(Import 1 μl of SABC in 99 μl of SABC Dilution Buffer.)
Analytical Procedures <br> The TMB substrate was equilibrated at 37 ° C for 30 minutes before the reagents were added to the wells. When diluting samples and reagents, they must be thoroughly mixed. It is recommended to draw a standard curve for each test.
1. Set standards, test samples and control (zero) holes on the pre-coated plates, respectively, and record their positions. It is recommended to measure each standard and sample in duplicate. Wash the plate 2 times before adding standard, sample and control (zero) holes!
2. Add sample and biotin detection antibody: Add 50 μL of standard each time, blank sample or sample is good. Add sample/standard dilution buffer to the blank wells. Immediately add 50 μL of biotin to detect antibody working solution to each well. Cover us with Plate Sealant. Gently tap the plate to ensure thorough mixing. Incubate for 45 minutes at 37 °C. (The solution is added to the bottom of the micro-ELISA plate to avoid the inner wall to do the best to stroke and foam.)
3. Washing: Aspirate each well and wash, repeat the process three times with each fill for cleaning and use a spray bottle, multi-channel pipette wash buffer (approximately 350 μL), manifold dispenser or automatic washer. Complete removal of liquid at each step is critical to good performance. After the last wash, remove the remaining wash buffer to inhale or pour. Turn the plate over and gently wipe the absorbent paper.
4. HRP-streptavidin conjugate (SABC): 100 [mu]L ABC working solution was added to each well. Cover the new Plate Sealer. Incubate for 30 minutes at 37 °C.
5. Washing: Repeat the aspirating/washing process five times.
6. TMB Substrate: Add 90 μL of TMB substrate to each well. Cover with a new plate sealant. Incubate at 37 ° C for about 15-20 minutes. Protected from light. The reaction time can be shortened or extended according to the actual color change, but not more than 30 minutes. The reaction can be terminated when a significant gradient appears in the standard wells.
7. Stop: Add 50 μL of Stop Solution to each well. The color immediately turns yellow. The order in which the stop solution is added should be the same as the matrix solution.
8. OD measurement: Use a to determine the optical density (OD value) of each well to be set to 450 nm. You should open the microplate reader in advance, preheat the instrument, and set the test parameters.
Calculation Results <br> Average repeat readings for each standard and sample. The best fit curve is drawn from the points on the graph by creating a standard curve to plot the average OD value of each standard on the y-axis or x-axis versus concentration on the x-axis or y-axis. It is recommended to use some professional software to do this calculation, such as curve expert 1.3 or 1.4. In the software interface, the best fit equation for the calculated standard curve is used for the OD value and the concentration of the standard sample. The software will calculate the sample concentration after entering the OD sample. In addition, you can enter the corresponding fitting equation and OD value to add the sample to Excel to get the concentration sample. If the sample has been diluted, the concentration calculated from the standard curve must be multiplied by the dilution factor. If the OD of the sample exceeds the upper limit standard curve, it should be retested after proper dilution. The actual concentration is the calculated concentration multiplied by the dilution factor. It is recommended to use a professional software curve expert for 1.3. For details, please visit: http://
Note: If the measured sample is diluted, multiply the dilution factor by the interpolated concentration to obtain the pre-dilution concentration.
Summary
1. Wash the plate 2 times before adding the standard, sample and control (zero) holes!
2. Add 50 μL of standard or sample to each well.
3. Immediately add 50 μL of biotin detection antibody to each well.
4. Incubate for 45 minutes at 37 °C.
5. Pipette and wash the plate 3 times.
6. Add 100 μL of SABC working solution to each well. Incubate for 30 minutes at 37 °C.
7. Pipette and wash the plate 5 times.
8. Add 90 μL of LTMB substrate. Incubate at 37 ° C for 15-20 minutes.
9. Add 50 μL of Stop Solution. Read 450nm immediately.
10. Calculation of results
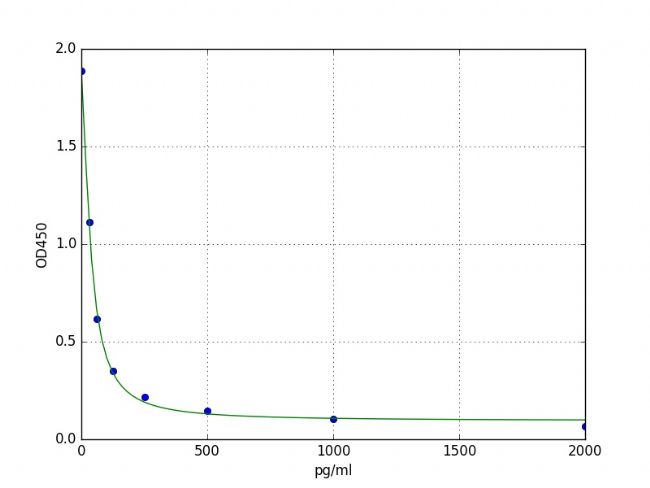
Typical data and standard curve
The results of typical standard procedures for the PGE2 ELISA kit are listed below. This standard curve is generated in our lab for demonstration purposes only. Users should obtain a standard curve according to their own experiments. (N/A=Not applicable)
| X | Pg/ml | 0 | 31.25 | 62.5 | 125 | 250 | 500 | 1000 | 2000 |
| Y | OD450 | 1.887 | 1.111 | 0.617 | 0.35 | 0.217 | 0.146 | 0.106 | 0.067 |
Specificity <br> This assay has high sensitivity and excellent specificity for the detection of PGE2. No significant cross-reactivity or interference between PGE2 and analogs was observed.
Note: Due to prior art and knowledge, it is difficult to perform cross-reactivity detection between PGE2 and all analogs, so cross-reactivity may still be present.
Resuscitation <br> The matrix listed below is spiked with a certain level of PGE2 and the recovery is calculated by comparing the measured value to the expected amount of PGE2 in the sample.
| Matrix | Recovery range (%) | Average(%) |
| Serum(n=5) | 89-103 | 95 |
| EDTA plasma(n=5) | 85-97 | 91 |
| Heparin plasma(n=5) | 86-104 | 93 |
Linearity <br> The linearity of the kit was determined by testing the incorporation of the appropriate concentration of PGE2 and its serially diluted sample. The results are demonstrated by calculating the concentration and the expected percentage.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| Serum(n=5) | 85-102% | 91-102% | 89-101% | 86-104% |
| EDTA plasma(n=5) | 85-99% | 94-100% | 82-98% | 84-101% |
| Heparin plasma(n=5) | 83-100% | 81-97% | 90-98% | 91-93% |
Precision <br> Determination of internal precision (accuracy within the measurement): Three samples with low, medium and high levels of Ig were tested 20 times on one plate, respectively.
Inter-assay precision (accuracy between measurements): Three samples with low, medium and high levels of Ig were tested on 3 different plates, 8 replicates per plate.
CV(%)= SD/meanX100
Within the batch: CV<8%
Inter-batch: CV<10%
stability
The stability of an ELISA kit is determined by the rate of loss of activity. Under appropriate storage conditions, the kit's failure rate is less than 10% on the expiration date.
| Standard (n=5) | 37°C for 1 month | 4°C for 6 months |
| Average(%) | 80 | 95-100 |
Reminder: In order to minimize the additional impact on performance, operating procedures and laboratory conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly recommended that the entire assay be performed by the same operator from start to finish.
Similar products
Human prostaglandin E2 (PGE2) ELISA kit   Literature citation
Mouse prostaglandin E2 (PGE2) ELISA kit
Porcine prostaglandin E2 (PGE2) ELISA kit
Rat prostaglandin E2 (PGE2) ELISA kit
Prostaglandin E2 (PGE2) ELISA Kit
Chlorhexidine Gauze Dressing,Chlorhexidine Gauze,Chlorhexidine Gauze Wound Dressing,Chlorhexidine Gauze Dressing for Burns
Roosin Medical Co.,Ltd , https://www.roosinmedical.com