Model NO.: LSI14
Pharmaceutical Technology: Chemical Synthesis
Drug Reg./Approval No.: Sfda:H20003688
Transport Package: as Your Request
Specification: 2ml: 0.1mg*10
Origin: China
HS Code: 2933330011
Model NO.: LSI14
Pharmaceutical Technology: Chemical Synthesis
Drug Reg./Approval No.: Sfda:H20003688
Transport Package: as Your Request
Specification: 2ml: 0.1mg*10
Origin: China
HS Code: 2933330011
Product name: fentanyl citrate injection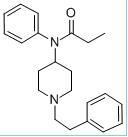
Specification: 10ml:0.5mg*2
The main components : fentanyl citrate, whose chemical name is N-[1- (2 - phenylethyl) 4-perazine] - n-phenylalanamide citrate.
The auxiliary materials are sodium chloride and injection water.
Traits : this product is colorless transparent liquid.
Indications : this product is a powerful analgesic medicine. It is applicable to the sedation and analgesia before, during and after anesthesia. It is commonly used in compound general anesthesia.
1.It is used to administer and induce anaesthesia before anesthesia, and is used for various operations as adjuvant medication and general anesthesia.
A mixture of 25mg and 0.05mg of Droperidol and 0.05mg of this product, before anesthesia, can make the patient quiet and indifferent to the environment, but can still cooperate.
2.Before, after and during the operation of all kinds of severe pain.
Usage dosage:
1.Adult intravenous injection.
The initial volume of the total hemp was 0.001 to 0.002mg/kg (with fentanyl meter, the same with fentanyl);
The main operation was 0.002-0.004 mg/kg;
In extracorporeal circulation heart surgery, the total amount of weight was calculated at 0.02~0.03 mg/kg, and the amount of maintenance can be divided into half of the initial amount every 30 to 60 minutes, or the continuous static drop, which is generally 0.001 to 0.002mg/kg per hour.
The total hemp was inhaled with nitrous oxide at the same time as weight 0.001-0.002 mg/kg;
The analgesic analgesia of the adjunct was 0.0015-0.002mg/kg as the adjuvant drug.
2.Analgesic before and after anesthesia for adult anesthesia: intravenous infusion of 0.0007~0.0015 mg/kg.
3.Children's pain: no regulation under 2 years old, 2-12 years old by weight 0.002~0.003 mg/kg.
4.Analgesic after adult operation: epidural administration, initial amount of 0.1 mg, sodium chloride injection diluted to 8ml, can be repeated every 2-4 hours, maintaining the amount of half of the initial amount each time.
Adverse reaction:
1.General adverse reactions are vertigo, blurred vision, nausea, vomiting, hypotension, sphincter spasm, laryngeal convulsion and sweating.
I have muscle twitches.
2.Severe side effects include respiratory depression, asphyxia, muscular rigidity and bradycardia, such as delayed treatment, respiratory arrest, circulatory inhibition and cardiac arrest.
3.Fentanyl citrate injection is addictive, but is lighter than pethidine.
Contraindications:
bronchial asthma, respiratory depression, hypersensitive patients of this product and myasthenia gravis.
It is forbidden to use with monoamine oxidase inhibitors (such as phenylhydrazine, pargyin and so on).
Notes:
1.This product is a national special management of narcotic drugs, must strictly abide by the management regulations of the state of narcotic drugs, the drug storage place of hospital and the hospital shall be locked, prescription color should be distinguished with other medicine prescription.
All levels of the custodian shall comply with the system of succession and shall not be negligent.
2.Fentanyl citrate injection must be on monoamine oxidase inhibiting drugs (e.g., furazolidone, c kappa hydrazine) stopped more than 14 days before delivery, and should try small doses (1/4 dosages), otherwise the unexpected happens, serious complications, clinical manifestations of sweat, muscle rigidity, blood pressure rise after the first dip, respiratory depression, cyanosis, coma, high fever, convulsions, and eventually to circulation collapse and death.
3.Cardiac arrhythmia, liver, kidney dysfunction, chronic obstructive pulmonary disease, respiratory reserve reduction and cerebral trauma coma, increased intracranial pressure, brain tumors and other patients with respiratory depression.
4.Fentanyl citrate injection has a certain irritation. It must not be strayed into the trachea or bronchi or applied to the skin and mucous membranes.
5.Epidural injection of this product analgesia, general work 4 ~ 10 minutes, 20 minutes of cerebrospinal fluid concentration peak, at the same time can have the whole body itch, role of 3.3 ~ 6.7 hours, and there is still a slow breathing rate and reduce the possibility of tidal volume, should be dealt with in a timely manner.
6.This product is not an intravenous anesthetic, although a large number of rapid intravenous injection can make the brain disappear, but the patient's stress response still exists, often with the knowledge of the procedure.
7.Quick injection can cause chest wall, abdominal wall muscle stiffness and influence ventilation.
8.The athlete should be careful.
Drug interactions:
 1.This product is similar to the pethidine chemical structure, and the two drugs can be cross-sensitive.
2.Fentanyl citrate injection and central depressants, such as hypnotic sedative, diazepam, etc.) (the barbiturates, antipsychotics (such as phenothiazine) and other narcotic analgesics and general anesthetics have synergy, such as share should be carefully and appropriately adjust the dosage.
3.This product is combined with 80% oxidized ammonia, which can induce the heart rate to slow down, the myocardial systolic decrease, the cardiac output decrease, and the left ventricular function is particularly obvious.
4.Muscle relaxant dosage can be due to the use of fentanyl citrate injection reduced correspondingly, and muscle relaxants can relieve muscle rigidity of the product, in case of apnea, duration is long, should identify the central (department) caused by the product use, or peripheral (due to muscle relaxant effect on neuromuscular junction N2 receptors).
5.Central inhibitors such as barbiturates, tranquilizers, anesthetics, and the effects of depression, such as combined application, should be reduced by 1/4 ~ 1/3.
SFDA:H20003688
This product is a prescription drug and must be purchased by prescription
Â
Â
 Product name: fentanyl citrate injection

Specification: 10ml:0.5mg*2
The main components : fentanyl citrate, whose chemical name is N-[1- (2 - phenylethyl) 4-perazine] - n-phenylalanamide citrate.
The auxiliary materials are sodium chloride and injection water.
Traits : this product is colorless transparent liquid.
Indications : this product is a powerful analgesic medicine. It is applicable to the sedation and analgesia before, during and after anesthesia. It is commonly used in compound general anesthesia.
1.It is used to administer and induce anaesthesia before anesthesia, and is used for various operations as adjuvant medication and general anesthesia.
A mixture of 25mg and 0.05mg of Droperidol and 0.05mg of this product, before anesthesia, can make the patient quiet and indifferent to the environment, but can still cooperate.
2.Before, after and during the operation of all kinds of severe pain.
Usage dosage:
1.Adult intravenous injection.
The initial volume of the total hemp was 0.001 to 0.002mg/kg (with fentanyl meter, the same with fentanyl);
The main operation was 0.002-0.004 mg/kg;
In extracorporeal circulation heart surgery, the total amount of weight was calculated at 0.02~0.03 mg/kg, and the amount of maintenance can be divided into half of the initial amount every 30 to 60 minutes, or the continuous static drop, which is generally 0.001 to 0.002mg/kg per hour.
The total hemp was inhaled with nitrous oxide at the same time as weight 0.001-0.002 mg/kg;
The analgesic analgesia of the adjunct was 0.0015-0.002mg/kg as the adjuvant drug.
2.Analgesic before and after anesthesia for adult anesthesia: intravenous infusion of 0.0007~0.0015 mg/kg.
3.Children's pain: no regulation under 2 years old, 2-12 years old by weight 0.002~0.003 mg/kg.
4.Analgesic after adult operation: epidural administration, initial amount of 0.1 mg, sodium chloride injection diluted to 8ml, can be repeated every 2-4 hours, maintaining the amount of half of the initial amount each time.
Adverse reaction:
1.General adverse reactions are vertigo, blurred vision, nausea, vomiting, hypotension, sphincter spasm, laryngeal convulsion and sweating.
I have muscle twitches.
2.Severe side effects include respiratory depression, asphyxia, muscular rigidity and bradycardia, such as delayed treatment, respiratory arrest, circulatory inhibition and cardiac arrest.
3.Fentanyl citrate injection is addictive, but is lighter than pethidine.
Contraindications:
bronchial asthma, respiratory depression, hypersensitive patients of this product and myasthenia gravis.
It is forbidden to use with monoamine oxidase inhibitors (such as phenylhydrazine, pargyin and so on).
Notes:
1.This product is a national special management of narcotic drugs, must strictly abide by the management regulations of the state of narcotic drugs, the drug storage place of hospital and the hospital shall be locked, prescription color should be distinguished with other medicine prescription.
All levels of the custodian shall comply with the system of succession and shall not be negligent.
2.Fentanyl citrate injection must be on monoamine oxidase inhibiting drugs (e.g., furazolidone, c kappa hydrazine) stopped more than 14 days before delivery, and should try small doses (1/4 dosages), otherwise the unexpected happens, serious complications, clinical manifestations of sweat, muscle rigidity, blood pressure rise after the first dip, respiratory depression, cyanosis, coma, high fever, convulsions, and eventually to circulation collapse and death.
3.Cardiac arrhythmia, liver, kidney dysfunction, chronic obstructive pulmonary disease, respiratory reserve reduction and cerebral trauma coma, increased intracranial pressure, brain tumors and other patients with respiratory depression.
4.Fentanyl citrate injection has a certain irritation. It must not be strayed into the trachea or bronchi or applied to the skin and mucous membranes.
5.Epidural injection of this product analgesia, general work 4 ~ 10 minutes, 20 minutes of cerebrospinal fluid concentration peak, at the same time can have the whole body itch, role of 3.3 ~ 6.7 hours, and there is still a slow breathing rate and reduce the possibility of tidal volume, should be dealt with in a timely manner.
6.This product is not an intravenous anesthetic, although a large number of rapid intravenous injection can make the brain disappear, but the patient's stress response still exists, often with the knowledge of the procedure.
7.Quick injection can cause chest wall, abdominal wall muscle stiffness and influence ventilation.
8.The athlete should be careful.
Drug interactions:
 1.This product is similar to the pethidine chemical structure, and the two drugs can be cross-sensitive.
2.Fentanyl citrate injection and central depressants, such as hypnotic sedative, diazepam, etc.) (the barbiturates, antipsychotics (such as phenothiazine) and other narcotic analgesics and general anesthetics have synergy, such as share should be carefully and appropriately adjust the dosage.
3.This product is combined with 80% oxidized ammonia, which can induce the heart rate to slow down, the myocardial systolic decrease, the cardiac output decrease, and the left ventricular function is particularly obvious.
4.Muscle relaxant dosage can be due to the use of fentanyl citrate injection reduced correspondingly, and muscle relaxants can relieve muscle rigidity of the product, in case of apnea, duration is long, should identify the central (department) caused by the product use, or peripheral (due to muscle relaxant effect on neuromuscular junction N2 receptors).
5.Central inhibitors such as barbiturates, tranquilizers, anesthetics, and the effects of depression, such as combined application, should be reduced by 1/4 ~ 1/3.
SFDA:H20003688
This product is a prescription drug and must be purchased by prescription
Â
Â
Â
Shenzhen Guangyang Zhongkang Technology Co., Ltd. , https://www.syztreatment.com