Jacquelyn Cole and Rui Chen
TharSFC, a Waters Company, Pittsburgh, Pennsylvania, USA.
introduction
When comparing multiple applications of HPLC and SFC, it was found that SFC provides better selectivity and shorter analysis time due to the low viscosity and high diffusivity of the supercritical liquid.
SFC can be used to reduce the cost significantly in the preparation mode because the fraction is typically collected in a smaller volume of volatile alcohol and thereby reduces considerable purification work. The continued shortage of acetonitrile (ACN) has also prompted multiple industries to evaluate whether SFC can replace acetonitrile-dependent reversed-phase liquid chromatography (RPLC) in analysis and purification. Now, scientists are exploring and companies are developing more detectors for SFC including UV, FID, MS and ELSD.
The Evaporative Light Scattering Detector (ELSD) was originally designed for high performance liquid chromatography (HPLC) for mass-based non-volatile compound detection. Because this detection mechanism does not depend on the optical properties of the compound, ELSD is considered to be a universal detector and is particularly useful for detecting analytes that do not have an ultraviolet chromophore. Many pharmaceutical and chemical laboratories use ELSD in conjunction with UV detection and mass spectrometry (MS) to ensure the “authenticity†of universal detection for chromatographic analysis of mixed or complex mixed compounds. ELSD has also been found to be widely used in the analysis and purification of natural products including dietary supplements, sports nutrition, vitamins, organic foods, beverages, cosmetics and beauty products.
We have reported a case study of the use of SFC ELSD for the analysis of sweeteners in sports drinks2 and a detailed evaluation of the SFC ELSD test parameters3, both of which were of analytical scale. Here we present a feasibility study on ELSD for small-scale preparative SFC. We hope that the results described here will enable professionals to incorporate ELSD into their daily workflows involving analytical and preparative scale SFCs.
test
material
Ketoprofen and acetaminophen were purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Ketoprofen and acetaminophen were dissolved in HPLC grade methanol to prepare a 0.5 mg/mL stock solution for analytical assays and a 5 mg/mL stock solution for purification. A silica gel column (analytical type: 4.6 x 250 mm, filled with 5 μm particles; preparative type: 10 x 250 mm, filled with 6 μm particles) was purchased from Princeton Chromatography Instruments (Princeton, NJ, USA).
spectrum analysis
Each test used an SFC Investigator II instrument equipped with a Waters 2998 Photodiode Array (PDA) detector and Waters ELSD (Milford, MA) (Waters subsidiary T harSFC, PA, USA) Pittsburgh).
The ELSD signal (analog signal output) is transmitted to the SuperChrom software via an analog to digital converter. A 10 μL loop was used for each analytical injection, and a 200 μL loop was used for each preparative injection. The ELSD uses a stainless steel tee with an inner diameter of 0.010 inches and a 25 μ x 50 cm PeekSil tube to split between the PDA and the back pressure regulator. The analytical method was run at a flow rate of 4 mL/min (85:15 CO2/methanol), a back pressure of 150 bar, and a temperature of 40 ̊C. The ELSD conditions for the analytical method are set as follows: gain = 1000, nebulizer temperature = 45 ̊ C, pipe temperature = 45 ̊ C, pressure = 30 psi. The preparative method was run at a flow rate of 10 mL/min (80:20 CO2/methanol), a back pressure of 150 bar, and a temperature of 40 ̊C. The ELSD conditions are set as follows: gain = 1000, nebulizer temperature = 28 ̊ C, pipe temperature = 50 ̊ C, pressure = 60 psi. The compensation pump on the collection device was set to pump 2 mL of methanol per minute. For purification applications, a continuous injection of 500 μL of injection five times, or 1 mg of each compound (2 mg of two compounds), the total injection of each compound is 5 mg (both compounds) 10 mg).
Results and discussion
Figure 1 shows an analytical SFC chromatogram of a mixture of ketoprofen/acetaminophen obtained using ELSD and UV. There is a slight time lag between the ELSD and the UV signal (about 2 seconds). Since ELSD signals are used for fraction triggering, lag time is especially important for preparative chromatography. Care should be taken to ensure that the signal processing time is consistent with the time the effluent reaches the collection valve, or that appropriate timing compensation is required to minimize losses during collection. For example, a 25 μ × 50 cm PeekSil tube was used in our experiments to split the main flow into the ELSD. Small inner diameter tubes are preferred to minimize flow into the ELSD and minimize sample loss. However, the length of the tube is determined by the time lag between the UV and ELSD signals. Longer tubes will further reduce the amount of sample flowing into the ELSD due to increased resistance; however, this will also increase the time lag between the two signals. In this example, the 2 second time lag is less than 3% of the total peak width.
Second, we optimized the ELSD settings for preparative injections. The results are shown in Figure 2. There have been two changes from analytical to preparative settings: flow rate increased from 4 mL/min to 10 mL/min, and modifier percentage increased from 15% to 20%; these two changes will result in increased fluid flow into the ELSD. In our previous study 3, the effects of gas flow rate and evaporation temperature were described. In short, the higher the gas flow rate, the shorter the residence time, the fewer the number of scatterings, and the worse the signal. The higher the gas flow rate, the more small particles are formed during the spraying process. The reduction in particle size also results in a decrease in the intensity of the scattered light. In addition, it is also recommended to maintain the evaporation temperature at as low a level as possible to maintain particle size uniformity and to obtain a better signal by enhancing the dissolution crystallization. This is consistent with the results shown in Figure 2: Increasing the gas flow rate and lowering the evaporation temperature appear to produce a narrower peak with fewer "spikes". Note that the sample concentration for preparative injections is much higher; sensitivity is no longer the primary concern. Quite the contrary, the goal of optimization is to produce reliable and reproducible signals. A representative preparative SFC chromatogram is shown in Figure 3. Figure 3(b) shows the SFC chromatogram of the superimposed injection. The cycle time is further shortened by superimposing the injection, further increasing the production capacity. Recovery and Purity were determined by re-injecting the collected fractions and comparing the resulting peak area to a pre-rendered calibration curve. In this study, the recoveries of ketoprofen and acetaminophen were 88% and 84%, respectively, and the purity was 98% and 100%, respectively.

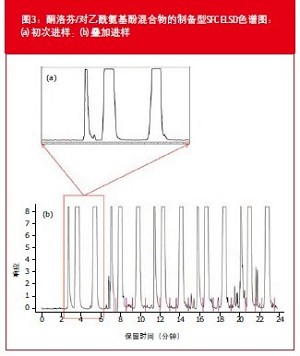
in conclusion
In this study, we demonstrate that the use of ELSD can trigger fraction collection for small-scale preparative SFCs. The recovery rate of the standard used in this study is greater than 84%, and the purity is greater than 98%. The fundamental principles of instrument configuration and ELSD optimization should apply equally to large-scale preparative SFCs.
references
[1] M. Dreux, M. Lafosse and L. Morin-Allory, Liquid Chromatography and Gas Phase
Chromatography International Edition, 9, 148–156 (1996).
[2] JL Lefler and R. Chen, Liquid Chromatography and Gas Chromatography
Edition, Applied Anthology Supplement, 26(6), 42–43 (2008).
[3] T. DePhillipo, JL Lefler, and R. Chen, Liquid Chromatography and Gas Chromatography
Spectrum European Edition, Applied Anthology Supplement, 22(3), 38-39 (2009).
About Waters Corporation ( )
For more than 50 years, Waters Corporation (NYSE: WAT) has made significant advances in medical services, environmental management, food safety and global water quality monitoring by providing practical and sustainable innovations that have created business for laboratory-related organizations. Advantage.
As a pioneer in a range of separation science, laboratory information management, mass spectrometry and thermal analysis technologies, Waters technology's breakthroughs and laboratory solutions create a lasting platform for customer success.
With $1.64 billion in revenue and 5,400 employees in 2010, Waters will continue to lead customers around the world to explore science and achieve excellence.
Adipic Acid
Adipic acid is mainly used in producing nylon fiber and engineering plastics, and polyurethane in quantity. Most of parts are used to produce PU, liquid for sole, polyurethane foam, etc. A small number of them are used for high quality lubricating oil.
High quality ester of adipic acid can be used as PVC and its copolymer, plasticizer of natural synthetic rubber. A large number of adipic acid ester is used to produce food packaging, thin film and PVC for insulation.
Adipic acid is also used for sour agent of food and drink. It is not easy to deliquesce, so it is used in dry food, and can be kept in a long time.

Adipic Acid 124-04-9,Adipic Acid,Adipic Acid Raw Material,99.7%Min Adipic Acid,Fine Adipic Acid
Shandong Tiancheng Chemical Co., Ltd. , https://www.akdchemical.nl


