Small molecule sickle cell disease drugs gain FDA breakthrough drug status
January 10, 2018 Source: US source of medicine: Lu Renbing
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];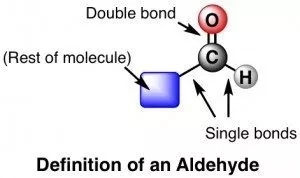
news
Today, biotechnology company Global Blood Therapeutics, Inc. (GBT) announced that its small molecule sickle cell disease (SCD) drug voxelotor (formerly known as GBT440) has achieved FDA breakthrough drug status, based on two phase II clinical and voxelotor A partial phase of clinical data. It is said that this is the first time that SCD drugs have obtained FDA breakthrough drug status, and GBT shares have risen by 20% today. Voxelotor was previously eligible for FDA approval, orphan drug, orphan drug for children, and was also eligible for EMA's priority drug.
Drug source analysis
Sickle cell disease is a blood disease caused by hemoglobin mutations. There are 100,000 and 60,000 patients in the United States and Europe, and 300,000 babies worldwide suffer from this disease every year. Patients in developed countries can live to 50-60 years old through blood transfusions and sound health care, while African patients with higher incidences die before the age of five. The mutant hemoglobin is easily polymerized after dissociation from the oxygen molecule, causing the red blood cells to be sickle-shaped and die faster. These red blood cells have a lifespan of 16-21 days, which is much lower than normal red blood cells for 120 days, causing anemia. These death sickle red blood cells also block capillaries, causing pain and organ failure.
Because SCD is caused by variant genes and is suitable for gene therapy, Bluebird Bio's LengiGlobin is best known. A patient with this drug was almost cured, and the industry once had great hopes for this product. However, the latter patients were not as effective as the first one. When the ASH annual meeting announced this result, the stock fell 11%. Last year, Emmaus Life Sciences' antioxidant L-glutamine powder (trade name Endari) was approved to control the symptoms of patients with sickle cell disease (SCD) over 5 years of age, and became a small molecule drug that relieves SCD symptoms after hydroxyurea.
Voxelotor is a small molecule drug containing an aldehyde group that reversibly forms a covalent bond with hemoglobin, thereby changing the conformation of the protein and reducing its polymerization rate. So voxelotor is a drug that changes the course of the disease, not just the symptoms. Drugs containing aldehyde groups are rare, and only one topical ophthalmic drug, alcaftadine, is marketed in addition to several macrolide antibiotics. In general medicinal chemistry optimization, the aldehyde group should be replaced by other groups as much as possible, because the aldehyde group is very active and inconsistent, it will cause serious toxicity. Formaldehyde is a well-known toxic substance, and an article published in a natural journal last week claimed that acetaldehyde, an alcohol metabolite, can cause stem cell variability and cause cancer. Voxelotor has data showing that it is well tolerated, with two patients sympathizing with the use of voxelotor, but allegedly not related to Voxelotor.
GBT is only 20% up when it gets a BTD. Investors seem to be a bit over-excited. After all, this is not an approved listing. Many BTD drugs are not available for listing, and there are examples of BTD eligibility. The stock market reaction may be related to the expectation of small molecule SCD drugs. Although gene therapy is more accurate, it is also very expensive and the side effects are difficult to reverse. Small molecule drugs are easy to use and relatively inexpensive, especially after the patent expires, so it is a good thing for both the patient and the payment system. If Voxelotor is listed as the first oral small molecule drug containing aldehyde groups, it is a minor breakthrough in the history of medicinal chemistry.
  US-Chinese medicine source website:
Tetanus Vaccine,Hepatitis B Vaccine For Adults,Tetanus Booster,Td Vaccine
FOSHAN PHARMA CO., LTD. , https://www.forepharm.com