Medical Network January 18th Recently, domestic 3 drug companies have been approved for clinical trials of nine-valent cervical cancer vaccine, 2022 cervical cancer vaccine may be listed, this time China will be synchronized with the world.
â–Chinese multi-drug enterprises obtained clinical trial license for 9- valent cervical cancer vaccine
On January 16, Watson Biotech announced that the "recombinant human papillomavirus nine-valent virus-like particle vaccine" independently developed by its controlling subsidiary Zerun Bio was received by the CFDA on January 15, 2018. Approvals.
November 2017, by the Xiamen University, diagnostic reagents and vaccines Engineering Research Center and Church Health Limited's Xiamen sea Biotechnology Co., Ltd. Cosmos National Infectious Diseases, Beijing Tso Bio-Pharmaceutical Co., Ltd. jointly developed the nine-valent cervix The cancer vaccine was approved by the CFDA for clinical trials.
This is the second, the world's second, China's first nine-valent cervical cancer vaccine approved for clinical trials, except for the nine-price cervical cancer vaccine approved by Merck in 2015.
In the same period, Bowei Bio's recombinant human papillomavirus nine-valent vaccine (Hanson's yeast) and Merck's nine-valent cervical cancer vaccine Gardasil 9 also obtained permission to conduct clinical trials in China.
â–GSK , Merck's cervical cancer vaccine market is highly competitive
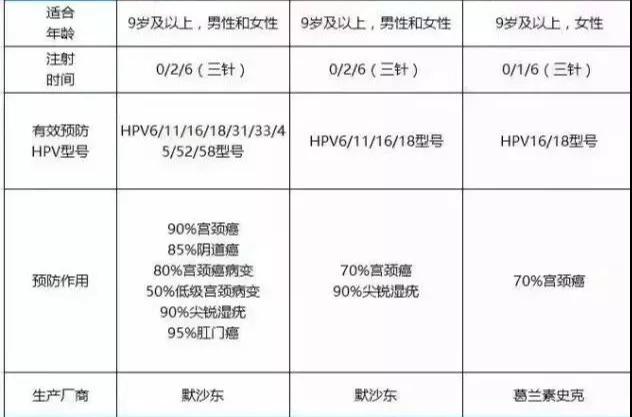
At present, there are three foreign vaccines that have been successfully developed to prevent HPV infection and cervical cancer, including GlaxoSmithKline's 2-valent HPV (16/18) vaccine and Merck's 4-valent HPV (6/11/16/18). Vaccine and 9-valent HPV (6/11/16/18/31/33/45/52/58) vaccine.
(Source: Network)
Both the above-mentioned 2-valent HPV vaccine and 4-valent HPV vaccine were approved for import registration and sold in China in 2017. The 9-valent HPV vaccine currently has no products on the market.
For the second consecutive year, 2015 and 2016, the global sales of HPV vaccines exceeded US$2 billion, making it the second largest vaccine in the world in terms of sales ranking after the 13-valent pneumococcal conjugate vaccine.
For a long time, Merck and GlaxoSmithKline have separated the major markets for global cervical cancer vaccines. Both have been competitors in this field. However, from the perspective of sales in the global market, Merck's “Jia Da Xiu†has always been ahead of GlaxoSmithKline's “Hirschâ€.
At first, the gap between the two sales was not too big, but as time went by, Jiada Xiu’s sales increased steadily, and the market share of “Cirrus†has been declining. According to the data, the annual sales volume of “Jia Da Xiu†in 2010 was 2.6 times that of “Xi Rui Shiâ€, and by 2015 the gap between the two sides has expanded to 14 times.
â– China is expected to launch a nine-valent cervical cancer vaccine simultaneously with the world
“Price†represents the type of virus segmentation covered by the vaccine. The higher the price, the larger the coverage. The 9-valent HPV vaccine developed by Watson Bio is an upgraded product of the 2-valent HPV vaccine, which is mainly used to prevent cervical cancer, vulvar cancer, and vagina caused by HPV16, 18, 31, 33, 45, 52, 58 infections in women of appropriate age. Cancer, anal cancer, and genital warts caused by HPV type 6 and type 11 infections, as well as for the prevention of anal cancer caused by HPV 16, 18, 31, 33, 45, 52, 58 infections in men of the appropriate age and by HPV type 6, 11 Genital warts caused by infection.
Some insiders said that in the future, China's cervical cancer vaccine market will be dominated by four-price and nine-price. If domestic HPV vaccine developers can form an impact price advantage in the 9th-order vaccine, the future market may be nine. The price is mainly.
For the nine-valent cervical cancer vaccine has been approved for clinical trials, some analysts pointed out that because clinical trials generally last four to five years, the nine-valent cervical cancer vaccine is expected to be listed in 2022. This time, China is expected to enjoy the nine-valent cervical cancer vaccine in sync with the world.
At the "National Health Industry Enterprise Management Association Drug and Medical Device Medical Transformation Branch" held on January 14, some people in the industry told Cypress that after China joined the ICH, it is expected to realize the multi-country recognition of a country's research data. More timely introduction of innovative drugs to the market, Chinese consumers will also be able to use new drugs globally.
Cultivating the cervical cancer vaccine market, it has a long way to go
Although the potential is huge, the current marketing situation is not optimistic. According to relevant media reports, cervical cancer vaccines are cold in Beijing, Weihai, Qianjiang and Rizhao. In Weihai, 2,200 bivalent cervical cancer vaccines were inoculated only 152 after one month of marketing. After the first batch of 900 bivalent cervical cancer vaccines in Qianjiang City were put on the market for half a month, no one was vaccinated; 17 vaccination clinics in Rizhao City were vaccinated. The maximum number is less than 50.
Some analysts believe that the domestic consumer vaccination awareness is weak, medical institutions are not active and other factors lead to cervical cancer vaccine cold. Cybertron has not yet received a response to the GSK on this matter.
In addition, affected by the previous vaccine incident, the country has tightened the control of the vaccine market. All vaccines are collectively purchased by the Chinese Center for Disease Control and Prevention (CDC) and then distributed to the provinces. It is unlikely that the CDC will enter the market when the vaccine enters the market. Large quantities of purchases.
It seems that although China has a potential consumer market of 500 million, the mine remains to be tapped. This is undoubtedly both an opportunity and a challenge for Chinese and foreign pharmaceutical companies.
Medical Isolation Shoe Covers,Isolation Shoe Boot Covers,Non Slip Shoe Boot Cover,Medical Isolation Shoe Boot Covers