Clinical progress of domestic breast cancer drugs
November 22, 2017 Source: Sina medicine Author: HEROIV
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];According to data published in China Cancer Incidence and Death 2003-2007 and China Cancer, the trends of standardized breast cancer incidence and mortality in China from 2003 to 2012 are as follows: Chinese women's breast cancer from 2003 to 2008 The incidence increased from 21.17/100,000 to 26.26/100,000, an increase of 17.65%. From 2010 to 2012, the incidence rate rose to 30.43/100,000, an increase of 43.74% compared with 2003. The average annual change in the incidence of breast cancer among women between 2003 and 2012 was 3.55%. The incidence and mortality of breast cancer in China's cancer-registered areas have generally increased gradually, and the burden of breast cancer in China has become increasingly severe. This article focuses on the clinical progress of HER2-positive breast cancer.
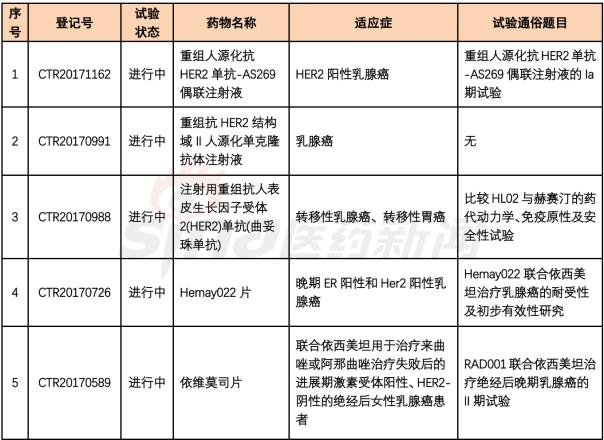
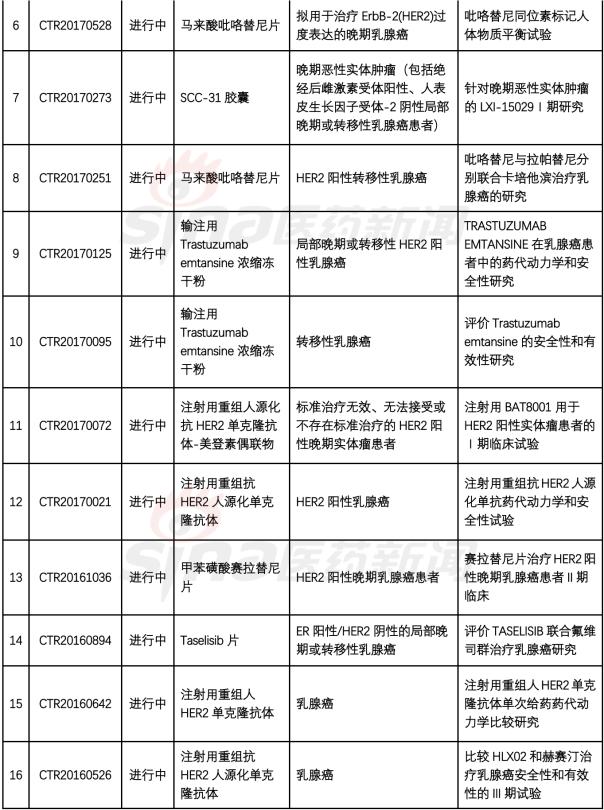
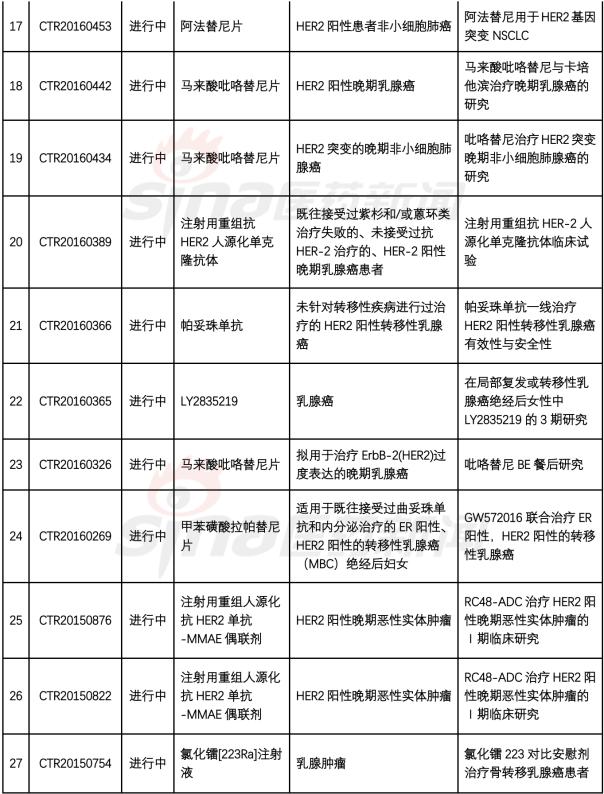
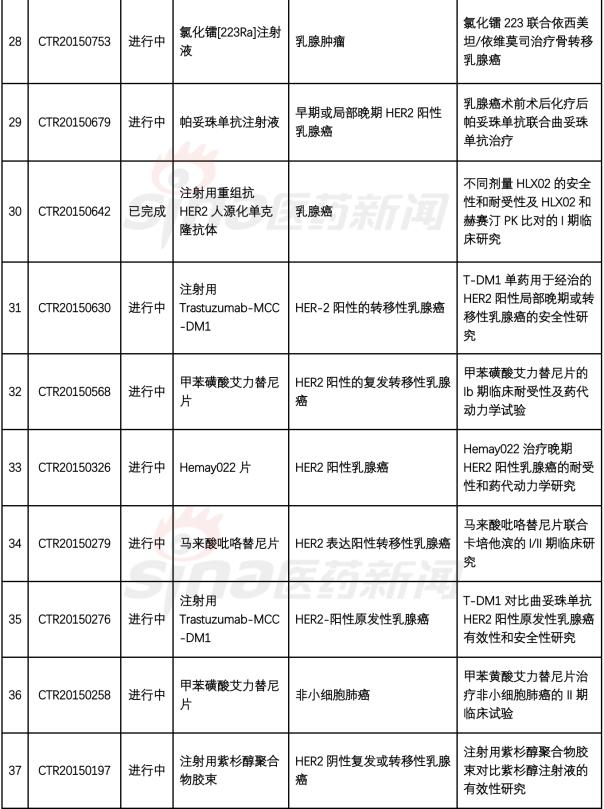
1. Recombinant humanized anti-HER2 mAb-AS269 coupling injection
Application for Enterprise / Organization: Zhejiang Pharmaceutical
A single-center, open-dose, escalating Phase Ia clinical study evaluated the safety, tolerability, and pharmacokinetic profile of ARX788 monotherapy for HER2-positive advanced breast cancer. This trial is intended to identify recombinant humanized anti-HER2 mAb-AS269 conjugates in HER2 positive advanced breast cancer subjects with HER2 results as immunohistochemistry (IHC) 3+ and/or fluorescence in situ hybridization (FISH)+ The maximum tolerated dose (MTD) / phase II recommended dose (RP2D) of the injection.
2. Recombinant anti-HER2 domain II humanized monoclonal antibody injection
Application for Enterprise / Organization: Livumab
A single-center, open-label, dose escalation Phase I clinical trial evaluated the safety and tolerability and pharmacokinetic profile of LZM005 injection in patients with advanced breast cancer. To evaluate the dose-limiting toxic DLT of LZM005 injection and confirm its maximum tolerated dose MTD for the subsequent phase II clinical trial. Recommended clinical dose evaluation. Safety and tolerability characteristics of LZM005 injection. Study on the pharmacokinetics of LZM005 injection. Characteristics; To study the immunogenicity of LZM005 injection; to evaluate the initial efficacy of LZM005 injection in advanced breast cancer.
3 , molecular EGFR / HER2 tyrosine kinase irreversible inhibitor Hemay022 combined with exemestane in the treatment of advanced ER- positive and Her2- positive breast cancer Ib clinical study
Application for Enterprise / Organization: Tianjin Hemei Bio
OBJECTIVE: To evaluate the safety of Hemay022 in combination with exemestane in patients with ER-positive and HER2-positive advanced breast cancer and the optimal tolerable dosing regimen for co-administration.
Secondary study objectives: To evaluate the pharmacokinetic profile of Hemay022 in combination with exemestane, and to evaluate the initial efficacy of Hemay022 in combination with exemestane; to explore the effect of ESR1 mutation on treatment outcome.
4. Phase II trial of RAD001 plus exemestane in the treatment of estrogen receptor-positive, HER-2 negative postmenopausal breast cancer with relapse or progression after treatment with letrozole or anastrozole
Application company / institution: Novartis Pharma Schweiz AG, Novartis Pharmaceuticals
5. Safety, Tolerance, Pharmacokinetic Characteristics and Anti-tumor Phase I Study of LXI-15029 in Patients with Advanced Malignant Solid Tumors in China
Advanced malignant solid tumors include postmenopausal estrogen receptor-positive, human epidermal growth factor receptor-2 negative locally advanced or metastatic breast cancer patients.
Application Enterprise / Organization: Shandong Luoxin Pharmaceutical Group Co., Ltd. / Shanghai Institute of Materia Medica, Chinese Academy of Sciences / Fudan University
6. Phase III clinical study of pyrrolidine maleate combined with capecitabine versus lapatinib plus capecitabine in the treatment of HER2- positive metastatic breast cancer
MAIN OBJECTIVE: The progression-free survival (PFS) of pyrrozine combined with capecitabine in the treatment of HER2-expressing metastatic breast cancer is superior to the lapatinib plus capecitabine regimen.
Application for Enterprise / Organization: Hengrui Medicine
7. Comparative study on the safety and efficacy of Trastuzumab emtansine and capecitabine lapatinib
Comparative evaluation of Trastuzumab emtansine and lapatinib + capecitabine for HER2-positive, unresectable locally advanced breast cancer (LABC) or metastatic breast cancer (MBC) previously treated with trastuzumab-based therapy Patient safety and effectiveness. Phase III clinical.
Applying for an enterprise / institution:
Roche Registration Limited/Roche Registration Limited
F.Hoffmann-La Roche Ltd./F.Hoffmann-La Roche Ltd.
Roche (China) Investment Co., Ltd. / Roche (China) Holdings Ltd.
8. A single-center, open, dose-increasing phase I clinical trial of BAT8001 for the safety, tolerability, and pharmacokinetics of HER2- positive solid tumors
Applying for an enterprise / institution: Biotech
Main research purposes: To evaluate the safety, tolerability, pharmacokinetic characteristics and immunogenicity of BAT8001 for injection.
Secondary study objective: To study the neutralizing antibodies and metabolites of BAT8001 for injection, and to evaluate the efficacy of BAT8001 for injection on HER2-positive solid tumors.
9. A multi-center phase II clinical trial of randomized open-parallel control in patients with HER2- positive advanced breast cancer treated with serratinib tosylate
Application for Enterprise / Organization: Qilu Pharmaceutical
The objective response rate (ORR) of 50 mg, 1000 mg combined with capecitabine and lapatinib combined with capecitabine in the treatment of HER2-positive advanced breast cancer.
10. Evaluation of TASELISIB in combination with fulvestrant for the treatment of a late-stage or metastatic breast during or after treatment with aromatase inhibitors
Taselisib tablets, phase III. Efficacy, safety, pharmacology, and self-reporting of postmenopausal women who are estrogen receptor-positive and HER2-negative, locally advanced or metastatic breast cancer, and who have undergone aromatase inhibitor treatment or disease relapse or disease progression after treatment Result (PRO).
11. Randomized, double-blind, multicenter, phase III clinical trial of recombinant anti-HER-2 humanized monoclonal antibody (GB221 ) or placebo plus capecitabine for the treatment of HER-2 positive advanced breast cancer
Application for Enterprise / Organization: Jiahe Bio
OBJECTIVE: To compare the progression-free survival (PFS) of the combination of GB221/capecitabine tablets versus placebo/capecitabine tablets.
Secondary study objectives: (1) Objective remission rate (ORR) from treatment period to week 12 (2) Time to disease progression (TTP) (3) Overall survival (OS) (4) Safety (5) Immunogenicity ( Anti-drug antibodies) (6) Subjects continued PFS during the treatment phase.
12. Phase III study comparing the efficacy and safety of pertuzumab versus placebo with Herceptin + docetaxel in the first-line treatment of HER2- positive metastatic breast cancer
Roche (China) Investment Co., Ltd. compared the PFS of the investigator and compared the combination of Herbine plus docetaxel with placebo plus Herceptin plus docetaxel in the treatment of newly diagnosed HER2 positive metastatic The efficacy of breast cancer (MBC) patients and the safety profile of the two treatment groups.
13, after the hormone receptor positive breast cancer in postmenopausal women HER2 negative by comparison with NSAI LY2835219 / Placebo and Comparative Fulvestrant associated with three research LY2835219 / Placebo of
Applying for an enterprise / institution: American Lilly
OBJECTIVE: To compare differences in PFS in patients with HR+, HER2-local recurrence or metastatic breast cancer in postmenopausal women compared with Abemaciclib in combination with NSAI (anastrozole or letrozole) versus placebo in combination with NSAI .
Key secondary objective: To compare differences in PFS in patients with HR+, HER2-local recurrence or postmenopausal women with metastatic breast cancer compared with vesevoscin versus placebo in combination with placebo.
14 , GW572016 combined with Herceptin plus AI treatment of Herceptin and endocrine therapy ER- positive, HER2- positive MBC postmenopausal subjects safety and effectiveness of phase III clinical
Application for Enterprise / Organization: Beijing Novartis Pharmaceuticals
MAIN OBJECTIVE: To verify that GW572016/trastuzumab/aromatase inhibitor combination therapy (treatment group A) is superior to trastuzumab/aromatase inhibitor combination therapy in overall survival (treatment group B) . Recruitment is completed.
15. Open-label, phase- dose phase I clinical study of RC48-ADC in the treatment of HER2- positive advanced malignant solid tumors with safety, tolerability and pharmacokinetics
Recombinant humanized anti-HER2 monoclonal antibody-MMAE coupling agent for injection from Yantai Rongchang Biological.
OBJECTIVE: To determine the maximum tolerated dose (MTD) and safety of RC48-ADC in patients with HER2-positive advanced malignant solid tumors, and to determine the recommended dose for phase II clinical trials.
OBJECTIVE: To investigate the pharmacokinetics (PK) characteristics of RC48-ADC in patients with HER2-positive advanced malignant solid tumors, and to observe the clinical efficacy of RC48-ADC in the treatment of HER2-positive advanced malignant solid tumors. Recruitment.
16, 223 radium chloride joint exemestane / everolimus treatment of HER2-negative breast cancer patients with bone metastasis II, randomized, double-blind, placebo-controlled trial
Application for Enterprise / Organization: Bayer Health Care
The aim of this study was to evaluate the use of radium dichloride-223 in combination with exemestane and everolimus for the treatment of human epidermal growth factor receptor 2 (HER2) negative, hormone receptor positive, and bone metastatic breast cancer subjects. Effectiveness and security.
17. Safety Tolerance, Efficacy and Pharmacokinetics of Emritinib Toluene Tablets Combined with Capecitabine in Patients with HER2- Positive Recurrent Metastatic Breast Cancer
Application for Enterprise / Organization: Shanghai Alice Medical
In clinical recruitment, stage I.
18 , paclitaxel polymer micelles for injection compared with paclitaxel injection first-line or second-line treatment of HER2- negative recurrent or metastatic breast cancer multi-center, open, randomized clinical trial
Application Enterprise / Organization: Wuxi Langci Biotechnology Co., Ltd.
OBJECTIVE: To evaluate the clinical efficacy of paclitaxel polymer micelles for injection as a first-line or second-line chemotherapy in patients with HER2-negative recurrent or metastatic breast cancer.
Secondary study objectives: To observe the safety of paclitaxel polymer micelles for injection in patients with HER2-negative recurrent or metastatic breast cancer.
Phase II, recruitment.
19. Efficacy and safety of Olaparib in patients with gBRCA1/2 mutation and high-risk HER2- negative primary breast cancer
OBJECTIVE: To evaluate the effect of adjuvant drug therapy on IDFS in patients with gBRCA1/2 mutations and high-risk HER2-negative primary breast cancer.
Secondary study objective: To evaluate the effect of adjuvant drug therapy on the incidence of OS, DDFS, new invasive primary breast cancer and/or new ovarian epithelial cancer, and PRO; to evaluate patients with harmful/suspected harmful mutations in the gBRCA gene Efficacy; evaluation of safety and tolerability. Exploratory purpose.
Phase III, recruitment.
20. A multicenter, randomized, double-blind, phase 3 study comparing Palbociclib with letrozole and placebo in combination with letrozole in the treatment of postmenopausal women with advanced breast cancer in Asian women .
Application for Enterprise / Organization: Pfizer
To demonstrate that in patients with ER(+)/HER2(-) Asian postmenopausal advanced breast cancer (ABC) who have not received any systemic anticancer therapy for advanced disease, Palbociclib in combination with letrozole is compared with placebo. Triazole can significantly prolong PFS. Recruitment is in progress.
21. A phase III randomized, double-blind, placebo-controlled trial comparing capecitabine plus sorafenib with capecitabine plus placebo in HER2- negative breast cancer
Solafenib (NEXA, BAY43-9006). Recruitment
Due to limited space, please visit the drug clinical trial registration and information disclosure platform for the latest progress.
(Original title: Has the clinical progress of domestic breast cancer drugs progressed?)
VTM sampling kits are designed for the collection, preservation, and transport of viruses, chlamydia, mycoplasma, and ureaplasma urea. They are now widely used for sampling and transporting prior to virus detection. MandeLab's VTM kits provide a safe and convenient way to collect and transport virus samples.
Nasal swab kit virus sampling tube,Used to collect samples of secretions from the throat or nasal cavity, Disposable
Jiangsu iiLO Biotechnology Co., Ltd. , https://www.sjiilogene.com