Medical Network June 28th, the latest drug report released by Ke Rui Wei An The Year's new drugs & biologics 2017 shows that in 2017, 113 new chemical and biological drugs were approved for the first time in the world. Among them, 52 new drugs have been on the market in 2017, including 7 first-in-class drugs; 36 are extended varieties (new indications, new compound combinations, new formulations of original products); 25 varieties have been approved but are not available for sale until December 31, 2017.
Anti-tumor drugs continue to be the hottest and most active areas of global drug discovery and marketing, and this trend is also reflected in the new drugs approved for marketing in China in 2017.
Among the 52 new drugs on the market, anti-tumor drugs accounted for the top 17 in the first place, accounting for about 33%, far more than the second central nervous system drugs (6, 12%). Among all 113 new drugs, there are 22 anti-tumor drugs, accounting for nearly 20%.
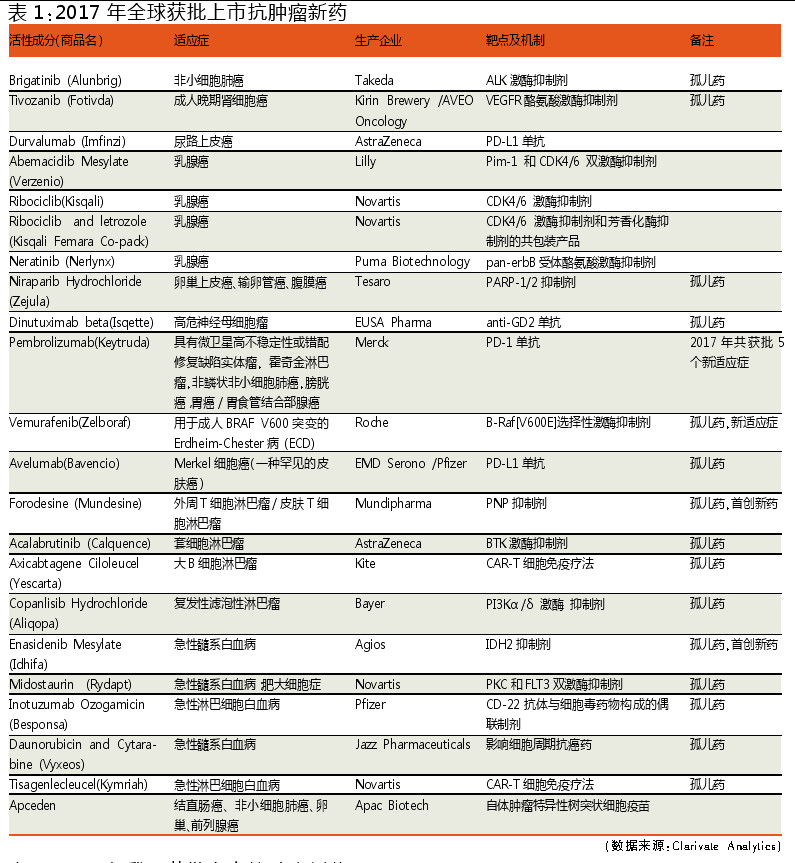
China's CDE "2017 Drug Review Report" shows that in 2017, CDE accepted the application for registration of domestically-produced Class 1 innovative drugs and the registration of new drug registration applications in the field of anti-tumor. In 2017, a total of 11 new anti-tumor drugs were approved for marketing in China, and 8 of them belonged to “new drugs with obvious clinical valueâ€. The priority review process was adopted to speed up the approval of the market.
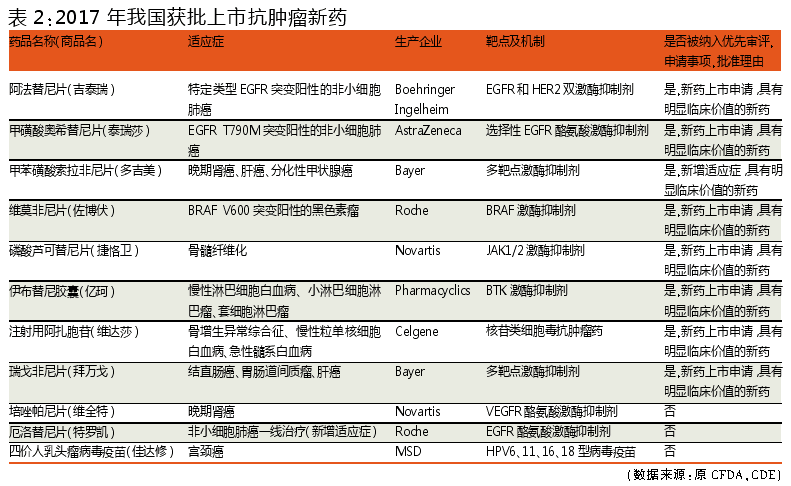
Compared with the new anti-tumor drugs approved in the world in 2017 and China, we can find many different points and similarities in this field at home and abroad, and we can also analyze the development trend of anti-tumor drugs in China.
Innovative target PK
The gap between China and foreign countries is still big, but China’s R&D is running fast.
Of the 22 new anti-tumor drugs approved worldwide in 2017, there are 10 different classes of kinase inhibitors (including 1 co-packaged product), 5 monoclonal antibodies (including 1 antibody-conjugated preparation), and 2 new targets. The first-in-class class of innovative drugs, two CAR-T cell immunotherapy drugs, one PARP-1/2 inhibitor, one cell cycle anti-tumor drug and one anti-tumor vaccine product.
In 2017, 11 new anticancer drug drugs approved in China include 9 different types of kinase inhibitors, 1 nucleoside cytotoxic antineoplastic agent and 1 anti-tumor vaccine product, among which kinase inhibitor anti-tumor drugs More than 80%. The 11 approved drugs are imported anti-tumor drugs that have been marketed abroad.
Indication PK
Different disease spectrum, Chinese listed products meet Chinese needs
The indications for the global approval of new anti-tumor drugs in 2017 are mainly concentrated in leukemia/lymphoma (9), breast cancer (4) and other rare diseases.
In the same period, China's approved anti-tumor drugs are mainly concentrated in lung cancer (3), leukemia/lymphoma/myelofibrosis (3), kidney cancer (2) and liver cancer (2).
The latest WHO report, World Cancer Report 2014, shows that there are about 14 million new cases of malignant tumors worldwide in 2012, and about 8.2 million deaths. It is estimated that by 2035, the number of new cases of malignant tumors worldwide will reach 24 million. The rapid increase in the incidence, mortality, disease burden and medication demand of malignant tumors worldwide has made anti-tumor drugs occupy a dominant position in the global pharmaceutical market. Of the top global best-selling drugs in 2017, six are anti-tumor drugs.
At present, the prevention and control situation of malignant tumors in China is also very serious. According to the latest data, there were about 3.084 million new cases of malignant tumors in China and 2.296 million deaths in 2014. There are about 21.8% of new cases of malignant tumors and 26.9% of the world. Malignant tumor deaths occur in China.
However, it is worth noting that the spectrum of malignant tumors in China is different from that in developed countries.
★ Major new malignant tumors in China: lung cancer (20.6%), gastric cancer (10.8%), colorectal cancer (9.75), liver cancer (9.6%), female breast cancer (7.3%), esophageal cancer (6.8%), thyroid cancer (4.5%), cervical cancer (2.7%), brain cancer (2.7%), and pancreatic cancer (2.4%). Lung cancer (20.6%), gastric cancer (10.8%), colorectal cancer (9.75), and liver cancer (9.6%) accounted for more than 50% of all new cases of malignant tumors in China.
★ Major new malignant tumors in North America: prostate cancer (14.6%), breast cancer (14.3%), lung cancer (13.4%), colorectal cancer (8.9%), bladder cancer (4.3%), melanoma (4.2%), Non-Hodgkin's lymphoma (3.9%), kidney cancer (3.6%), thyroid cancer (3.2%).
★ Major new malignant tumors in Europe: breast cancer (13.5%), colorectal cancer (13.0%), prostate cancer (12.1%), lung cancer (11.9%), bladder cancer (4.4%), gastric cancer (4.1%), kidney Cancer (3.3%), pancreatic cancer (3.0%), melanoma (2.9%).
The spectrum of different malignant tumors shows that there is a certain difference between the demand for clinical anti-tumor drugs in China and other countries, which in turn affects the types and market scale of specific listed drugs at home and abroad.
However, among the anti-tumor drugs approved for marketing in China in 2017, there are also several key drugs for rare tumors. It is expected that in the future, pharmaceutical companies will not be limited to common tumor types, but will start from the actual needs and market positioning of cancer patients in China, and introduce more rare tumor drugs to meet the treatment needs of cancer patients in China.
Drug review center PK
70% of the world's new drugs are orphan drugs, China attaches importance to clinical value
Of the 22 new anti-tumor drugs approved worldwide in 2017, 15 were granted FDA-approved orphan drug certification, and other drugs were supported by various countries' accelerated approval policies.
Of the 11 anti-cancer drugs approved for marketing in China in the same period, 8 are “new drugs with obvious clinical value†and then accelerated the approval of the market through the priority review process.
In order to improve the accessibility of patients, and encourage enterprises to accelerate the development of new drugs, it is a common practice in the world to give different support policies and measures to R&D enterprises in drug review and approval. The FDA has four special evaluation policies for accelerating the development of new drugs, such as fast-track identification, breakthrough therapy identification, accelerated approval and priority review. It also has long-term market exclusivity, tax reduction and R&D subsidies.
Since 2005, China has successively launched special approval, major special projects, special approvals, and priority review and approval to promote the accelerated drug review policy. In 2017, China joined ICH and became the eighth member of its global regulatory body, marking China's pharmaceutical products. Regulatory approvals are in line with international standards . In particular, the “Opinions on Deepening the Reform of the Examination and Approval System to Encourage the Innovation of Pharmaceutical Medical Devices †issued in 2017, “Opinions on Encouraging Drug Innovation to Implement Priority Evaluation and Approvalâ€, and the April 2018 State Council executive meeting on the import of anticancer drugs. The support policies for innovative drugs within the review process, mechanisms, clinical trials, and intellectual property rights have greatly benefited the R&D and listing of innovative drugs with obvious clinical value and urgent needs of patients in China.
Imported anti-tumor drugs continue to benefit from the in-depth reform of China's drug review and approval system: In 2017, the average time for the CFDA to approve clinical trials of imported anti-cancer drugs was 114 days, 129 days shorter than the 243 days in 2014; the approval of imported anti-cancer drugs The time to market was an average of 111 days, which was 309 days shorter than the 420 days in 2014. The approval of the new late-stage liver cancer drug Rigopheril imported in March 2017 was approved only 7 months later than the world's first listed country. . These figures indicate that there will be more imported anti-tumor drugs in the future to achieve simultaneous listing at home and abroad.
Breathing Filter,air filter,filter meshes,air breathe filter,breathing circuit filter
Anesthesia Medical Co., Ltd. , https://www.honestymed.com