On May 15th, Zhou Bin Research Group of the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences published the latest research results of “ Genetic Targeting of Organ-Specific Blood Vessels †in the international academic journal Circulation Research. This work establishes a new genetic operating system for more precise genetic targeting for gene knockout and overexpression.
Nanmo Biotech constructed a mouse model of sequential cross-genetic targeting tools for this study.
To date, genetic targeting tools have relied heavily on the Cre-LoxP homologous recombination system to address cell tracing problems for gene function studies. The accuracy of this system is highly dependent on the specificity of the promoter or gene expression that drives Cre. That is, if the Cre expression element is inserted into the A gene, Cre recombination can be exerted in the cell expressing the A gene. While the A gene is often not specifically expressed in a particular organ or tissue, this traditional approach has inherent limitations.
For example, the genetic tools commonly used in the study of blood vessels are VE-cad-Cre, Tie2-Cre, etc. However, these tools are labeled with all endothelial cells and cannot accurately study the blood vessels of specific tissues and organs. The blood vessels of different organs are heterogeneous in structure and function. The role of endothelial cells in different organs in the injury response is unique. Therefore, how to improve the accuracy of genetic targeting and develop tissue-specific Cre tool mice is of great significance for studying the in-depth functional study of specific cell types in a certain organ formation and regeneration process.
In this latest study, the researchers used the Cre-loxP and Dre-rox systems to perform sequential cross-genetic targeting manipulations with each other's recombination systems to accurately target organ-specific blood vessels.
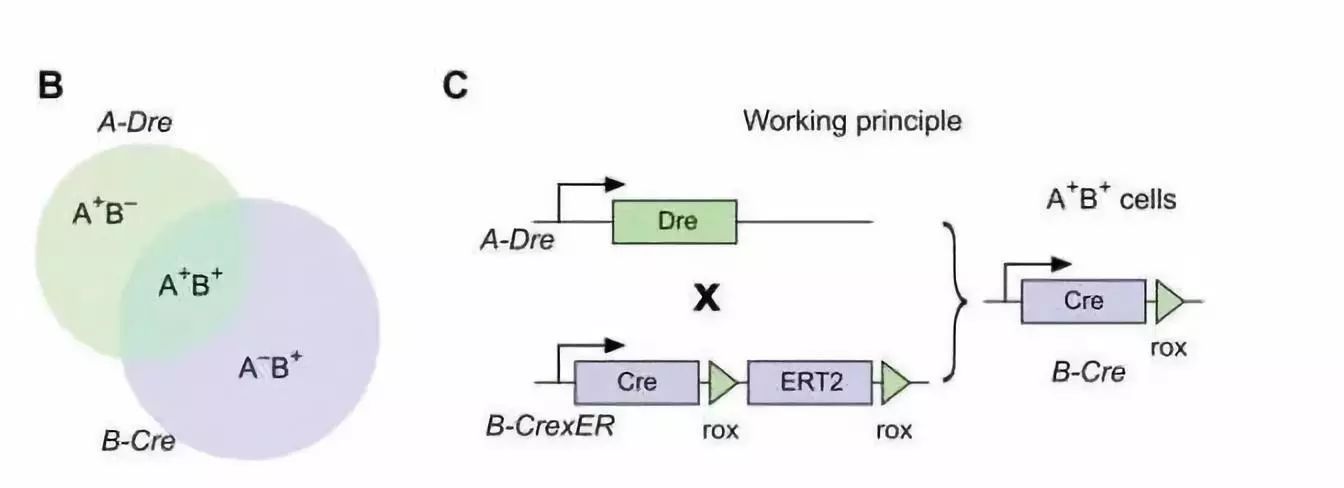
Core: Sequential Cross-Genetic Targeted Control System
Two elements:
Two different promoters drive Dre and Cre - A-Dre and B-CrexER (B) below.
A-Dre : Inserts Dre into the A locus by knockin, which makes the expression of Dre consistent with the expression profile of the A gene.
B-CrexER : Inserts the CrexER element into the B locus by knockin. CrexER is a novel inducible tool mouse that places two rox sites on either side of the estrogen receptor (ER), Cre-rox-ER-rox.
According to the design of the cross-targeting system in this order (Fig. C below), in general, the Cre-ER fusion protein stays in the cytoplasm and cannot enter the nucleus for the Cre-LoxP reaction. Only in cells expressing both the A gene and the B gene, the Dre recombinase recognizes and cleaves the Rox site, after which B-Cre is released from the cytoplasm into the nucleus for subsequent Cre-LoxP recombination.
Using the Nrg1-CrexER expressed by neural crest and the widely expressed CAG-Dre, it was verified that the Cre-rox fusion protein still has recombinase activity after excision of ER-rox by Dre, and specifically and effectively exerts Cre recombinase in vivo. effect. And CrexER itself has no leakage expression. These data validate the feasibility of Dre-mediated Cre expression strategy and confirm that this novel CrexER can be used for in vivo sequential cross-targeting.
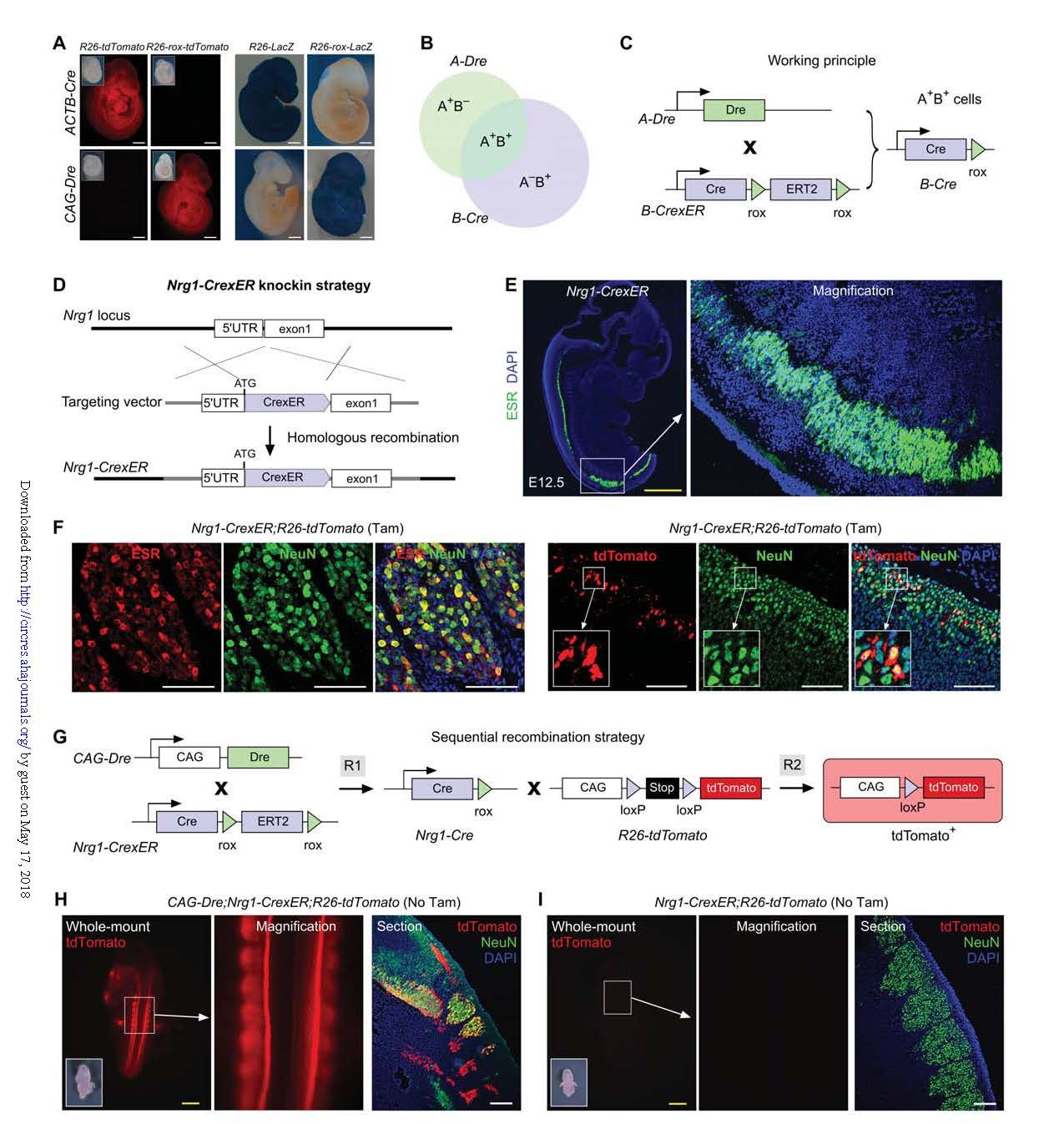
Figure 1. Proof-of-principle for a sequential recombination strategy for genetic targeting.
The sequential cross-genetic targeting manipulation system utilizes two promoters to more precisely define the labeled cells, and the final effective output is the Cre recombinase. The selection of the two marker genes A and B is critical to the success of the entire system, and requires a comprehensive understanding of the expression profiles in the previous literature and experiments.
In this study, the researchers found two Marker genes in the heart and brain, respectively, and successfully achieved specific labeling of vascular endothelial cells in the heart and brain.
Application 1: Cardiac Coronary Endothelial Cell Specific CoEC-Cre
Tie2-Dre: Tie2 is a pan-endothelial cell marker expressed in most blood vessels as well as certain bone marrow cell populations. Tie2-Dre tool mice were established by knocking in Dre into the ATG position of the endogenous Tie2 gene.
Wt1-CrexER: It has been reported that the mesothelial cell marker Wt1 is expressed in the developing coronary arteries, but not in other blood vessels. In addition to epicardial cells, Wt1-CreER labels coronary endothelial cells in the embryonic heart. The Wt1-CrexER tool mouse was established by inserting CrexER into the translation initiation codon (ATG) position of the Wt1 gene. (Fig. 2A-B)
Verification experiments showed that Tie2-Dre; Wt1-CrexER did indeed specifically and efficiently label coronary vessels (Fig. 2C-E). As an internal control, no tdTomato+ cells in Tie2-Dre; R26-tdTomato tissue were detected, thereby precluding Dre-loxP recombination. Wt1-CrexER; T26-tdTomato mice did not detect any tdTomato+ cells in the absence of tamoxifen treatment, indicating that Wt1-CrexER was not leaking.
These data indicate that Cre-loxP recombination occurs only in the Tie2-Dre and Wt1-CrexER dual-group systems, and Tie2-Dre; Wt1-CrexER (hereinafter referred to as CoEC-Cre) can exclusively specifically target cardiac blood vessels. Next, the application of the dual-group system in gene knockout or overexpression was verified.
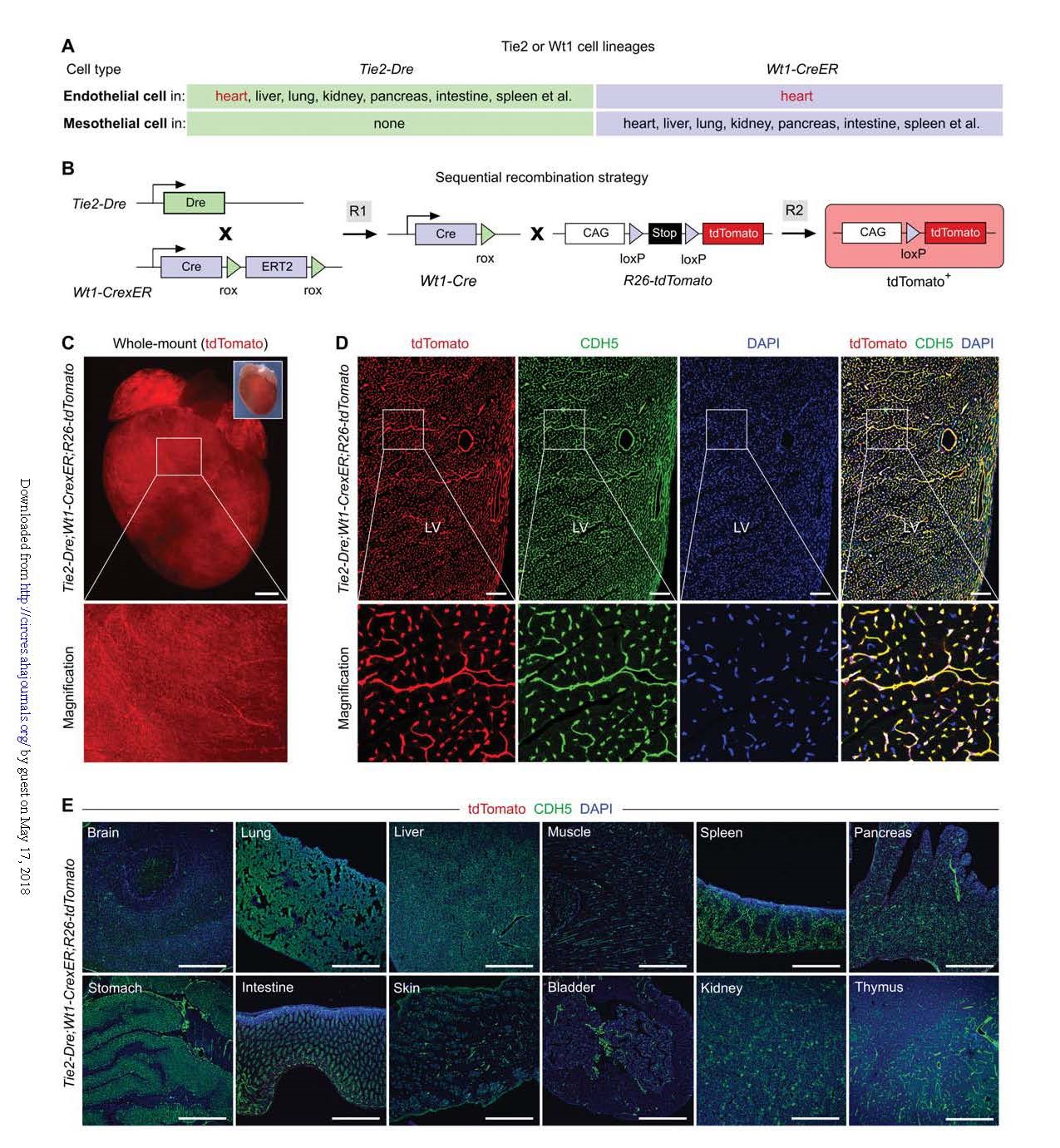
Figure 2. Genetic targeting of coronary vessels by Tie2-Dre; Wt1-CrexER line.
Specific gene manipulation in coronary vascular endothelial cells
Gene knockout in heart-specific blood vessels
VEGFR2 (Kdr) is ubiquitously expressed in endothelial cells of most organs. It is almost impossible to specifically target this gene in the coronary arteries. Therefore, the Kdr gene was selected as a target gene to verify the condition of the CoER-Cre system. Application in sex gene knockout.
Mating with CoEC-Cre (Tie2-Dre; Wt1-CrexER) by Kdr flox mice (Fig. 3A). In order to be able to trace at the same time, the R26-tdTomato reporter mouse was introduced.
Knockout group: Tie2-DRE; WT1-CrexER; Kdrflox/flox; R26-tdTomato mice;
The litter control group was: Tie2-DRE; WT1-CrexER; Kdrflox/+; R26-tdTomato mice.
E14.5 heart section immunostaining was used to detect tdTomato, VEGFR2 and CDH5, and the results showed that VEGFR2 was deleted in the dense myocardium of the mouse heart of the tdTomato+CDH5+ coronary endothelial cell knockout group (Fig. 3B-D). In the endocardial cells of myocardial trabeculae, VEGFR2 expression was not affected, indicating that CoEC-Cre recombination specifically targets Wt1-expressing coronary endothelial cells, but not endocardial endothelial cells (Wt1 is not expressed during embryogenesis).
Gene overexpression in cardiac-specific blood vessels
The human diphtheria toxin receptor (DTR) was selected as an overexpression gene. CoEC-Cre was crossed with R26-iDTR mice, where DTR expression was controlled by Cre-loxP recombination (Fig. 3E). Coronary endothelial cells expressing DTR can be specifically eliminated by diphtheria toxin (DT) treatment, thus verifying that this system is also feasible in gene overexpression. Immunostaining showed expression of DTR only in coronary endothelial cells (Fig. 3F); severe disruption of endothelial cell morphology, indicating apoptosis (Fig. 3G). Tie2-Dre; Wt1-CrexER; R26-iDTR mice There was a significant difference in the number of TUNEL+ cells in heart sections between the DT-administered group and the unadministered control group (Fig. 3H, I).
Combining the above two knockout and overexpression experiments, it is shown that the new gene targeting system can exclusively perform genetic manipulation in coronary endothelial cells without affecting other organ systems.
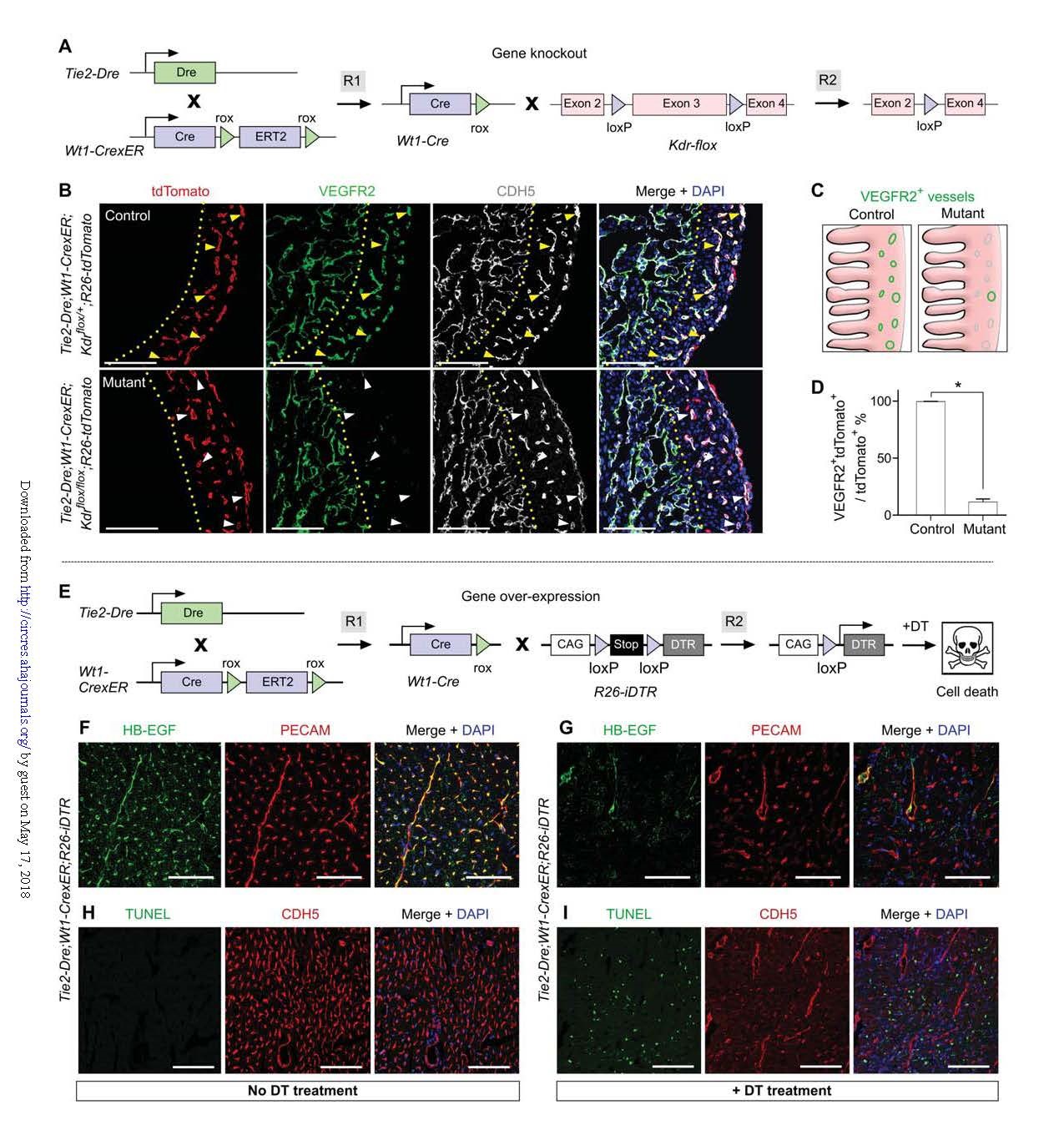
Figure 3. Gene knockout and over-expression in coronary endothelial cells.
Application 2: Brain endothelial cell specific BEC-Cre
Tie2-Dre: same as above
Mfsd2a-CrexER: Previous studies have shown that Mfsd2a is expressed in brain endothelial cells but also in other cell types (Fig. 4A). The cDNA encoding CrexER was inserted into the ATG position of the first exon of the Mfsd2a gene to establish a Mfsd2a-CrexER tool mouse, and the expression of CreER in the mouse was verified. Tie2-Dre tool mice were then mated with Tie2-Dre; Mfsd2a-CrexER double positive mice (hereinafter referred to as BEC-Cre) (Fig. 4B).
Compared to conventional Mfsd2a-CreER targeting cerebrovascular and neuronal, BEC-Cre is able to efficiently and specifically target cerebral blood vessels with specificity superior to Mfsd2a-CreER without labeling any neurons (Fig. 4C-E). More importantly, in addition to specificity, BEC-Cre is able to exert Cre recombination more effectively in brain endothelial cells than conventional Mfsd2a-CreER (Fig. 4E). In addition, BEC-Cre does not target any portal vein hepatocytes, nor does it target blood vessels in other organs or tissues. Percent quantitative statistics of tdTomato+ endothelial cells in brain and other organs or tissues also confirmed the efficiency and specificity of BEC-Cre mice (Fig. 4G).
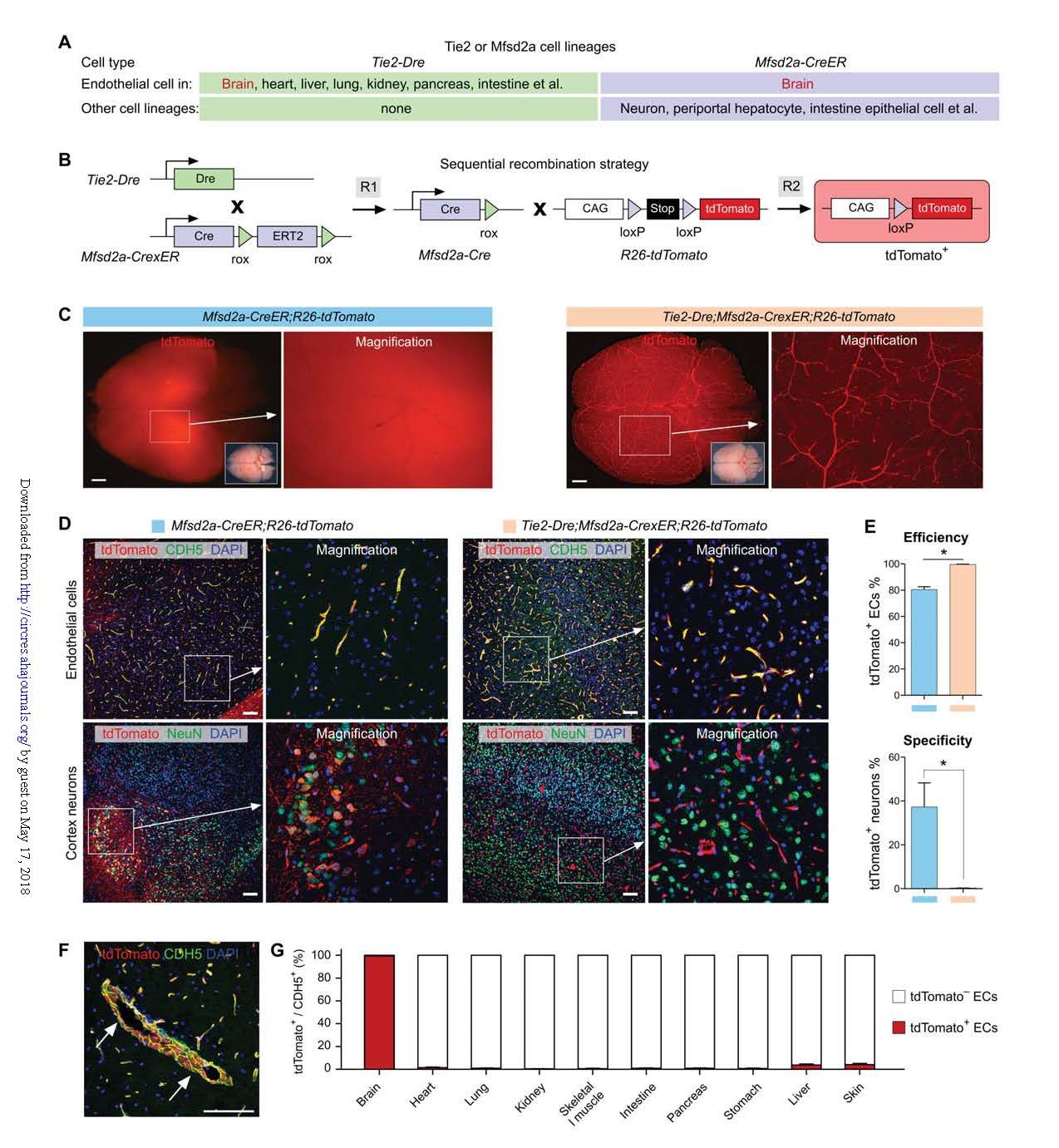
Figure 4. Specific and efficient brain endothelial cell targeting.
Gene manipulation in brain-specific vascular endothelial cells
Gene knockout in brain-specific blood vessels
Similarly, using VEGFR2 (Kdr) as a target gene again, a knockout group (Tie2-Dre; Mfsd2a-CrexER; Kdrflox/flox; R26-tdTomato) and a littermate control group (Tie2-Dre; Mfsd2a-CrexER; Kdrflox) were obtained. /+; R26-tdTomato) mice (Fig. 5A). In the neonatal mouse brain, the VEGFR2+ blood vessel was almost undetectable in the knockout group (Fig. 5B-C); but the decrease in VEGFR2 expression was not detected in the blood vessels of other organs or tissues, demonstrating the specific loss of Kdr in the cerebral blood vessels. . Compared with the control group, the vascular density in the knockout group was significantly reduced (Fig. 5D); the enlargement of the avascular region was also observed, and the cerebral hypoxia was significantly increased (Fig. 5E), confirming that the cerebral vascular density was reduced to tissue oxygen. The impact of the combination. In addition to reduced angiogenesis, knockout mice had abnormal morphological changes in the cerebral blood vessels (Fig. 5F).
In addition, by injection of 10-kDa dextran tracer assay and tdTomato, erythrocyte marker Ter-119, glucose transporter 1 (Glut1) immunostaining, capillary leaks were found, confirming the effect of Kdr deletion on vascular integrity (Fig. 6A) -C). Moreover, the protein composition of brain endothelial cells in the Kdr knockout group also changed, and the plasma membrane vesicle-associated protein (PLVAP) originally not expressed was significantly induced in BBB endothelial cells (Fig. 6D). In BEC-Cre-mediated Kdr-specific knockout mice, the expression of β-catenin (CTNNB1) was significantly reduced (Fig. 6E).
Taken together, these results, using BEC-Cre to mediate cerebrovascular specific knockout, circumvented early embryonic lethal death of conventional systemic Kdr knockout, and demonstrated that Kdr is required for central nervous system angiogenesis. The VEGF signaling pathway regulates genes involved in BBB endothelial cell integrity.
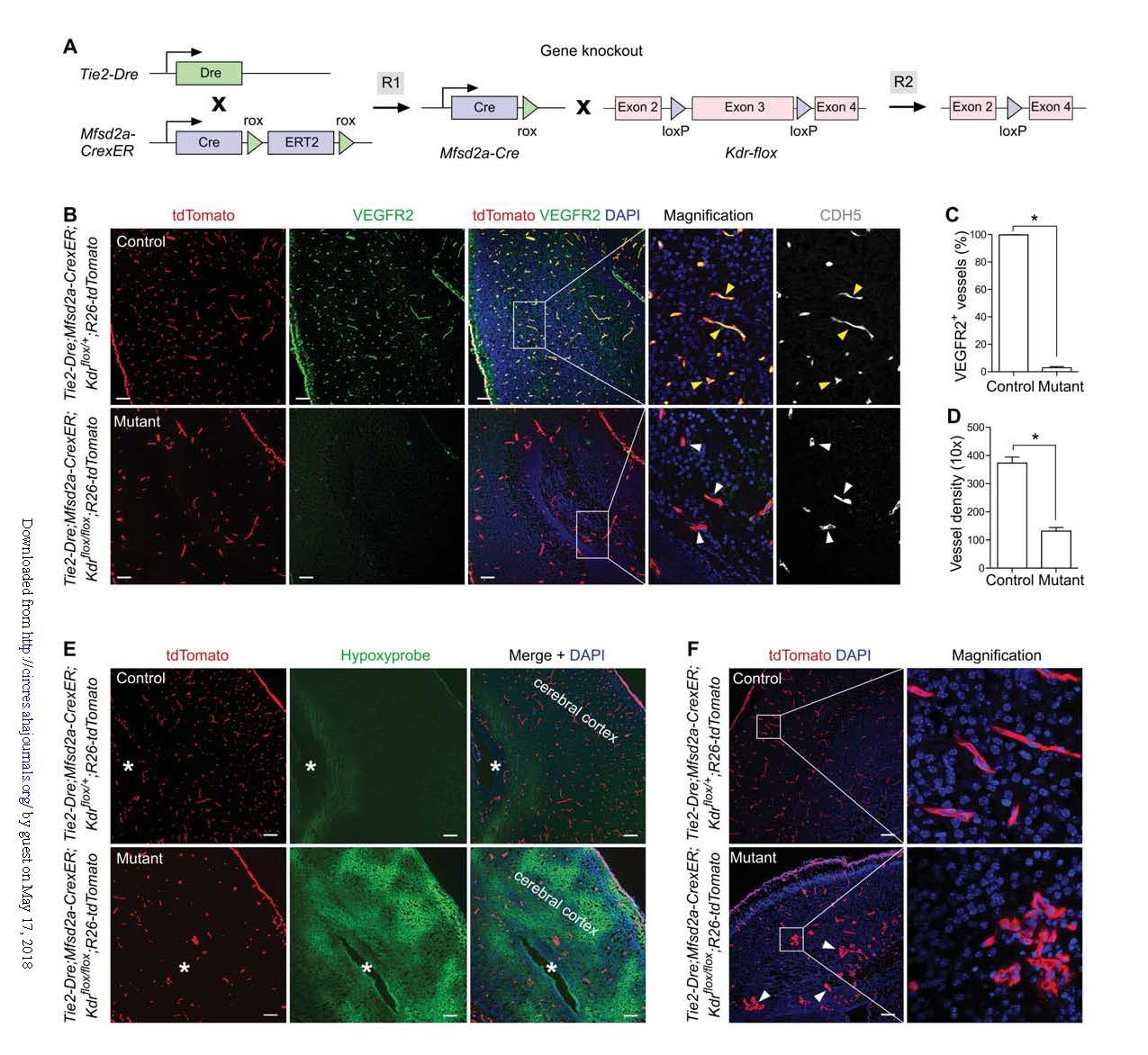
Figure 5. Specific knockout of VEGFR2 in brain endothelial cells.
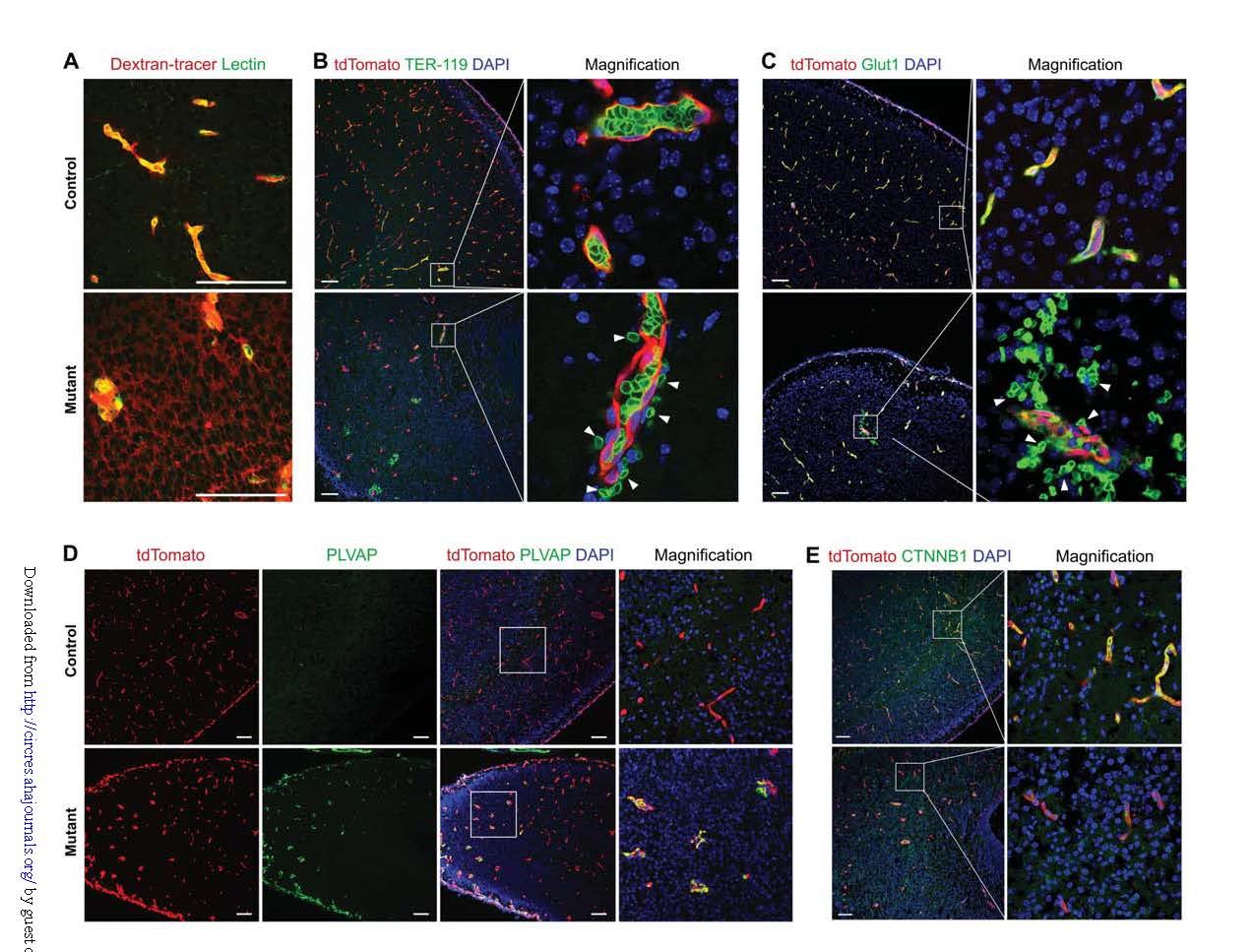
Figure 6. Vegfa-Vegfr2 signaling regulates BBB integrity.
This study successfully constructed cardiac coronary-specific Cre (CoEC-Cre) and cerebral vascular-specific Cre (BEC-Cre), and used a sequential cross-genetic targeting system to reveal that VEGF signaling pathway is involved in the regulation of cerebral angiogenesis and blood brain The formation of a barrier.
With the increasing popularity of knockin technology, the establishment of Cre tool mice has become more and more convenient; this makes the specific targeted gene knockout and gene overexpression more and more refined.
Dre's new sequential cross-genetic targeting system in combination with CrexER enables precise genetic manipulation in specific cell populations, providing an effective strategy for tissue-specific gene manipulation that can be applied to other biomedical applications. field. Is it wonderful?
Nanmo Biotechnology used the CRISPR gene editing technology and ES cell targeting technology to construct a multi-tool mouse model including Nrg1-CrexER, Nrg1-CreER, Wt1-CrexER, Tie2-Dre, Mfsd2a-CrexER.
If you have a good idea or inspiration, try it out and try it out carefully! We are willing to customize the service with a professional mouse model, and provide you with a better mouse model with the fastest speed to achieve scientific research dreams!
Article link
Urine Analyzer,Urine Analyzer For Lab,Lab Hospital Clinical Urine Analyzer,Urine Chemistry Analyzer
Guangdong Widinlsa International Co.Ltd , https://www.gdwidinlsa.com