
Morphological analysis of organotin compounds in biological tissues using GC/ICP-MS
introduction
Since organotin is widely used in agriculture, industry and households, its environmental impact has received great attention [1] . Butyltin and phenyltin have been widely used as active biocides, in antifouling coatings, polyvinyl chloride ( PVC ) stabilizers, wood processing, and other applications [2] . For example, when used in antifouling coatings, trace levels ( ppt ) of butyltin and phenyltin will cause severe damage to non-target organisms and accumulate in sediments and biota [3-4] . Therefore, the European Union ( EU ) included tributyltin ( TBT ) and its degradation products in the list of priority controlled pollutants (Revised Decision 2455/2001/EC of the Water Framework Directive 2000/60 /EC ) [4] . The US Environmental Protection Agency ( EPA ) has established environmental standards for aquatic aquatic organisms for TBT . The standard value for preventing marine aquatic organisms from chronic toxicity is 3 ng Sn/L , while the standard value for acute toxicity is 172 ng. Sn/L [4] . Since TBT , triphenyltin ( TPhT ) and their degradation products (disubstituted tin, monosubstituted tin and inorganic tin) have different toxicity and different environmental monitoring, it is necessary to The morphology is analyzed [5] .
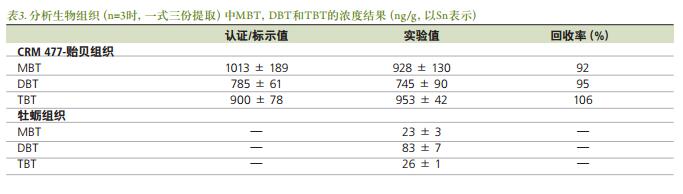
Nowadays, gas chromatography - inductively coupled plasma mass spectrometry ( GC/ICP-MS ) has the advantages of high sensitivity, good selectivity, and simultaneous analysis of various elements and multiple isotopes, thus becoming a trace analysis of metal organics. The preferred method [3] . Organotin morphology carried by GC / ICP-MS analysis in order to increase the volatility of each form needs to be added tetrapropylammonium sodium borate or sodium borate tetraethyl derivatization reaction. In addition, for the analysis of complex matrices, such as biological tissues and sediments, soft extraction is required prior to the derivatization of the organotin compound to maintain the morphology of the analyte unchanged. Open microwave extraction is the most popular extraction method due to its fast and efficient advantages [3] . This experiment used GC/ICP-MS ( CLARUS ® GC and NexION ® ICP-MS ) to analyze the form of organotin, more specifically, by external standard method for monobutyltin, dibutyltin and tributyltin in biological samples. analysis. Moreover, in the experiment, special attention was paid to the optimization of the GC transmission line parameters connecting GC and ICP-MS .

Experimental condition
Reagents and chemicals
Methyl monochloride monochloride ( MMT , 97 %), dimethyltin dichloride ( DMT , 97 %), trimethyltin chloride ( TMT , 97 %), monobutyltin trichloride ( MBT , 95 %), di-butyltin dichloride (DBT, 97%), and tributyltin chloride stock solution (TBT, 96%) were purchased from Sigma-Aldrich company â„¢, using glacial acetic acid (HAc) (GFS chemicals) Formulated. Derivatization was carried out using sodium tetrapropylborate (NaBPr 4 ) (Merseburger Spezialchemikalien, Germany ) . The buffer solution (0.1 M, pH 4.9) used in the derivatization process was prepared by dissolving sodium acetate and glacial acetic acid ( HAc ) in MQ water . HPLC grade isooctane and Optima grade methanol ( MeOH ) were purchased from Thermo Fisher ScientificTM .
High purity ammonium hydroxide ( NH 4 OH ), nitric acid ( HNO 3 ), hydrochloric acid ( HCl ) tetramethylammonium hydroxide ( TMAH , 25 %)
All were purchased from GFS Chemicals ( GFS Chemicals ). The standard reference material used in this experiment, the mussel tissue ( CRM477 ), was purchased from the Standard Reference Materials and Measurement Institute ( IRMM ; Kiel, Belgium), while the oyster tissue samples were from Arcachon Bay , France .
Instrumentation <br>The PerkinElmer ® Clarus 580 Gas Chromatography was coupled with a NexION 300D Inductively Coupled Plasma Mass Spectrometer ( ICP-MS ) for analytical analysis. GC and ICP-MS were coupled via a PerkinElmer GC/ICP-MS transmission line ( See Figure 1 ). It will be equipped with a capillary column deactivated within Silcosteel ® transmission line is inserted into the quartz tube torch and the central tube. The optimization of instrument operating conditions (eg, distance of inactivated capillary column, Ar gas composition, and oxygen ( O 2 ) flow rate) under dry plasma conditions was evaluated. An organo-tin was extracted from the matrix using a SP-D Discover focused microwave system ( CEM , Matthews, North Carolina, USA). The operating conditions of the instrument are shown in Table 1 . Data collection and analysis was performed using NexION ICP-MS and Chromera ® CDS software.
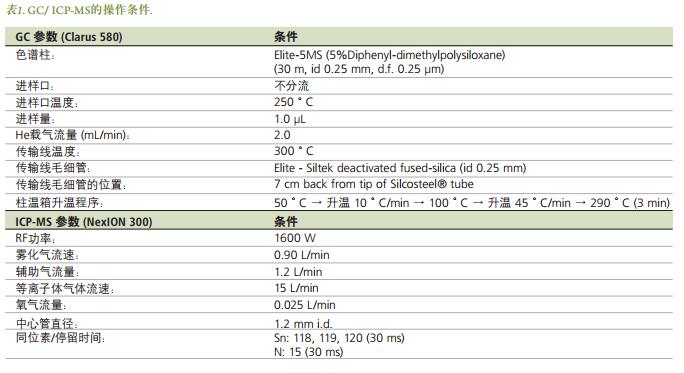
Sample Preparation
Sample preparation, microwave extraction and derivatization have been documented [3-4] . A stock solution of methyltin and butyltin ( 1000 μg / ml) was prepared using concentrated glacial acetic acid. The working solution was diluted in proportion with 1% HCl , prepared daily, and stored at low temperature. The standard series of standard solutions were repeatedly measured 3 times to obtain a calibration curve. For derivatization, about 4.4 g of sodium acetate was weighed , 1.0 ml of acetic acid was added , and then dissolved in MQ water and made up to 500 mL , and made into an acetate buffer solution (0.1 M, pH 4.9) . In addition, the use of MQ water, 2.5% NaBPr 4 formulated as a solution of derivatizing agent, and in the dark place.
Microwave extraction
In the microwave extraction of organotin compounds in mussel and oyster tissues, 0.15 g of dry sample was added to a 10 mL extraction vial , respectively , and then 4 mL of 25% TMAH was added to the dried sample . A small magnetic stir bar was added to each extraction vial to ensure adequate homogenization of the slurry during the extraction process. After microwave digestion, the sample can be centrifuged as needed, and the supernatant is transferred to a clean vial and stored in the refrigerator for testing. The microwave program used in this experiment is shown in Table 2.

Derivative
Derivatization was carried out in a 20 mL glass vial. First, the acetate buffer were added to 5mL is added to 20, 50, 100 and 200 μ L standard working solution containing 5 ng / mL of organotin. The pH of each solution was adjusted to 5.0 using HCl or NH4OH as needed . Was then added 1mL of isooctane and 250 μ L 2.5% of 4 NaBPr vial, capped bottle, and manually shaken for 5 minutes. The organic phase was then transferred to a GC vial for analysis. Three blank solutions were prepared in the same manner , on the one hand the detection limit determination can be carried out, on the other hand any contamination characteristics that may occur during sample preparation can be found. For the analysis of biological tissue, respectively, 30 μ L of 300 μ L mussel tissue and oyster tissue extracts added 5 ml of buffer, using HCl or NH 4 OH pH value of the solution was adjusted to 5.0. Then, were added to 1mL vial isooctane and 500 μ L of 2.5% NaBPr, capped bottle, and manually shaken for 5 minutes. The organic phase was transferred to a GC vial for analysis. Figure 2 summarizes the flow of the preparation of this experimental sample.
Working under clean conditions is very important for the success of low concentration analysis. The containers were cleaned with detergent and then soaked with nitric acid and hydrochloric acid ( 10 % V : V , MQ water). After soaking with different acids, rinse with MQ water. After the cleaning is completed, the container is placed under a laminar flow hood and stored for later use.
Results and discussion
Optimization of operating parameters mainly involves GC conveyor line and ICP-MS torch, such as internal capillary column distance, Ar composition gas flow rate (ie atomizing gas) and oxygen ( O 2 ), respectively, using carrier gas helium ( He ) Nitrogen ( N 2 ) impurities (isotopes 15 ) were evaluated for continuous signals. The addition of O 2 is necessary to prevent isooctane from producing carbon deposits in the ICP-MS sampling cone [5] . Although this experiment was optimized using a response value of 15 N + , a previous report using a helium / argon mixture ( 50 ppm ) was reported [6] .
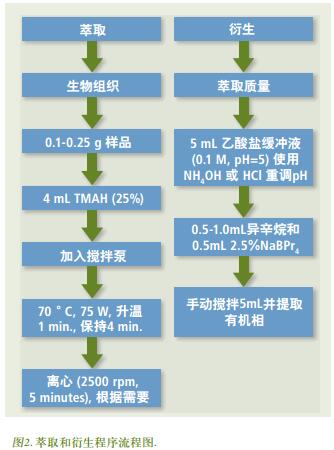
GC transmission line PerkinElmer provided, or may be used CLARUS AutoSystem ™ GC, it may also be used CLARUS or ICP-MS with the platform AutoSystem ™ GC NexION ELAN ® or hyphenated. The CLARUS GC system controls the temperature of the GC transfer line to maintain the analyte in the transfer line in the gas phase and prevent cold spots [7] . The Ar gas flow rate is controlled by an ICP-MS atomizer and heated prior to entering the transfer line. Sufficient flow is important for efficient delivery of the material to be tested into the plasma. Oxygen is fed through a tee (" T "), and oxygen is also passed through the transmission line. After completion of the set-point instrument torch 15 N + measured response value, and optimized for the 15 N + response. According to the previous work, the optimum value of O 2 gas addition and internal deactivation capillary column position ( relative position of the end of the Silcosteel ® tube in the transmission line ) were 0.025 L/min and 7 cm [7], respectively . By changing the Ar composition gas ( O 2 flow rate 0.025 L/min ) gas flow rate in the range of 0.8-1.0 L/min , when the 5 N + response value is observed to be the largest, the optimum Ar composition gas flow rate is determined. After determining the optimal conditions, an organic solvent (for example, isooctane, hexane, acetone, etc.) is injected to ensure that the substance to be tested can enter the plasma. Figure 3 is a typical plot of the 5 N + isotope signal when isooctane is injected . The inhibition of the 5 N + baseline signal at 2.5 minutes corresponds to the combustion of the organic solvent in the plasma, thus demonstrating the penetration of the GC gas stream into the plasma. The optimal conditions for the GC/ICP-MS system are listed in Table 1 .
Analytical performance
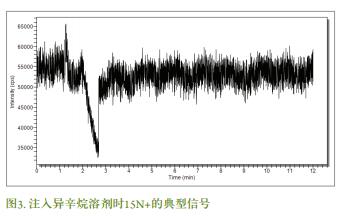
Analytical response characteristics were determined using a butyl tin standard solution. The formation of the calibration curve for the Sn isotope ( 118 , 119, and 120 ) was obtained by repeating three injections of 1 μL of the standard solution, and the concentration of the standard solution ranged from 0 (ie, blank) to 2 ng/mL , and was prepared using an isooctane solution. It can be seen from the 120 Sn response function (Fig. 4 ) that the calibration curve has good linearity and a satisfactory correlation coefficient ( R 2 value). Limit of detection (LOD = 3 σ blank / m ) obtained by each of the calibration response function, and 120Sn of MBT, DBT and TBT absolute detection limit of 18, 200 and 7 fg, respectively. The higher absolute detection limit of DBT may be due to the higher blank paper, which is related to the contamination caused by the containers and reagents used in the chemical preparation process. This phenomenon is very common because DBT is used in industrial synthetic plastics.
Chromatographic separation and method validation
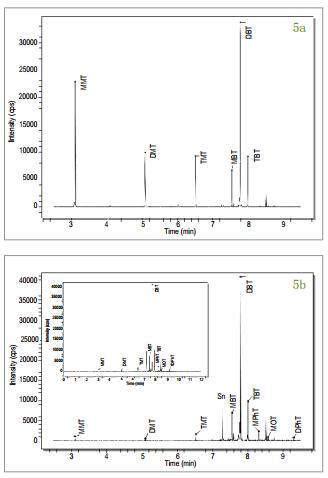
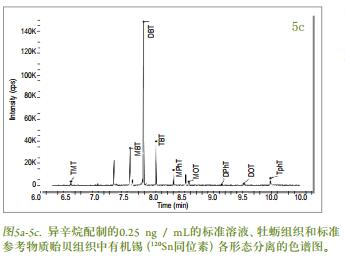
Figures 5a-c show chromatograms of the separation of organotin ( 120 Sn isotope) from 0.25 ng / mL standard solution, oyster tissue and standard reference material mussel tissue, prepared with isooctane . Multiple Sn isotopes ( 118 , 119 , 120 ) were monitored during the development of the method , but for the sake of simplicity, only 120 Sn results were listed . The chromatographic conditions (Table 1 ) were chosen primarily in such a way that the morphological species were separated from the solvent eluent interference zone [1] . The quantitative results of the butyltin compounds present in the biological tissue samples were obtained by an external standard method. The standard reference material for the mussel tissue extracted in triplicate was used for the validation of this experimental method. The experimentally measured MBT , DBT and TBT values ​​were compared with the certified values ​​and the results are shown in Table 3 . Although the concentration value of the butyl tin form substance in the oyster tissue sample was not calibrated, the experimental concentration value can be seen from Fig. 3 . The temperature program listed in Table 1 provides a guarantee for efficient separation of the tin-based compound, which can be separated in less than 11 minutes with a peak width of 2-3 s . Figure 5a is a chromatogram of a standard solution containing methyltin, butyltin species (mass 120 ) and a concentration of 0.25 ng / mL in isooctane under optimized conditions . From Fig. 5b and Fig. 5c , it can be seen that the chromatographic separation diagrams of oysters and mussels have other organic tin species commonly found in environmental samples, such as: octyltin, dioctyltin, monophenyltin, diphenyl. Base tin, triphenyltin, etc. All peaks in the chromatogram are clearly identified. Occur when 8.5 minutes peak may be due to impurities in NaBEt, NaBEt 4 is added in stage derivatization reagent, such that these impurities may form an organotin species generating ethylated product. Further work is needed to confirm these substances.
in conclusion
This paper presents a method for analyzing biological tissues using GC transmission lines to connect CLARUS GC and NexION ICP-MS detection system results. The morphology of butyltin has a good linear response function. After the parameter optimization is completed, the instrument LODs are determined . GC chromatographic separation of organotin compounds was applied to the analysis of standard reference materials for mussel tissue and oyster tissue. The external standard method was used to quantify and verify the morphology of butyl tin in biological tissues. The recovery rate obtained in this experiment was ≥92 %, and the relative standard deviation was ≤14 %.
references
1. Aguerre, S., Pécheyran, C., Lespes, G., Krupp, E., Donard, OFX, Potin-Gautier, MJ Anal. At. Spectrom. 16, (2001), 1429.
2. De Carvalho Oliviera, R., Erthal Santelli, R., Talanta 82 (2010), 9.
3. Monperrus, M., RodrÃguez Martin-Doimeadios, RC, Scancar, J., Amouroux, D., Donard, OFX Anal. Chem. 75, (2003), 4095.
4. Jiménez Moreno, M., Pacheco-Arjona, J., RodrÃguez González, P., Preud'Homme, H., Amouroux, D., Donard, OFX J. Mass Spectrom. 41, (2006), 1491.
5. Rodriguez Pereiro, I., Schmitt, VO, Szpunar, J., Donard, OFX, Lobinski, R. Anal. Chem. 68, (1996), 4135.
6. Castro, J., Tessier, E., Neubauer K., and Donard, OFX, “Mercury speciation in biological tissues and sediments by GC-ICPMSâ€, PerkinElmer Application Notes (), 2012.
7. Bouyssiere, B., Szpunar, J., Lobinski, R., Spectrochimica Acta Part B. 57, (2002), 805.
Fruits And Vegetables,Fresh Fruits And Vegetables,Fresh Fruit And Veggies,Fresh Vegetables
Xi'an Gawen Biotechnology Co., Ltd , https://www.agolyn-bio.com