According to the China New Drug Development Monitoring Database (CPM), the pharmaceutical company's new drug for ovarian cancer, Nila Pali, was accepted by CDE.
Niraparib is a once-a-day oral PARP inhibitor marketed under the trade name Zejula, developed by Tesaro for the treatment of ovarian and breast cancer.
Nilapali is the world's third marketed PARP inhibitor product and the world's first approved PARP inhibitor for all platinum-sensitive recurrent ovarian cancer patients regardless of BRCA mutation.
Nilapali was approved by the FDA on March 27, 2017 and approved by the EMA on November 16, 2017 for maintenance therapy in women with recurrent epithelial ovarian cancer, fallopian tube cancer, or primary peritoneal cancer.
On June 29, 2016, Tesaro released Phase III clinical data of Nilapa for advanced ovarian cancer recurring after chemotherapy. The results showed that ovarian cancer with BRCA mutation was orally administered once a day, with a median progression-free survival of 21 months, while the control group was only 5.5 months.
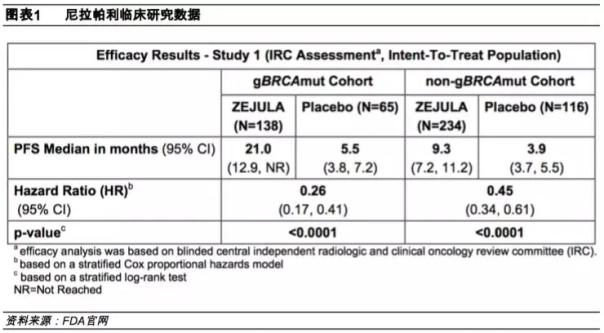
After seeing such exciting clinical results, Ding Ding introduced Nilapali to China at an appropriate time. On September 29, 2016, Ding Pharmaceutical entered into a strategic cooperation agreement with Tesaro, and Ding Pharmaceutical obtained the exclusive R&D and sales rights of Nira Pali in the Chinese market. It was approved in Hong Kong on October 22, 2018 for the treatment of ovarian cancer. It is suitable for patients with remission after platinum-containing chemotherapy, as a maintenance treatment to prevent the deterioration of the disease, and does not require BRCA or other biomarker testing.
According to the China New Drug Development Monitoring Database (CPM), there are currently 67 PARP inhibitors under research worldwide. Among them, there are three listed varieties, namely Astraco's Olapali, Clovis's Luca Pali and Tesaro's Nira Pali.
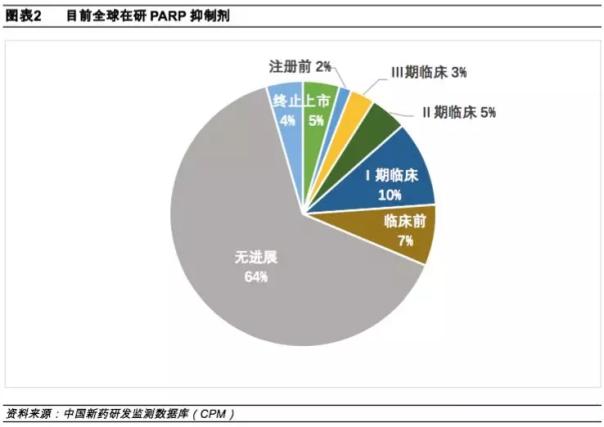
In addition to Nilapali, which is reported by Ding Ding Pharmaceutical, the companies currently deploying PARP inhibitors in China mainly include Baekje Shenzhou, Hengrui, Chuangnuo Medicine and Renfu Medicine.
The compliance of the drug, nilapali, which is taken only once a day, is particularly good. For patients with unknown BRCA mutations, the most clinically valid evidence for nilapali is strong. Therefore, regardless of whether the patient has BRCA mutations, nilapali is preferred as second-line maintenance therapy.
Have to sigh: the eyes of Ding Ding are really good!
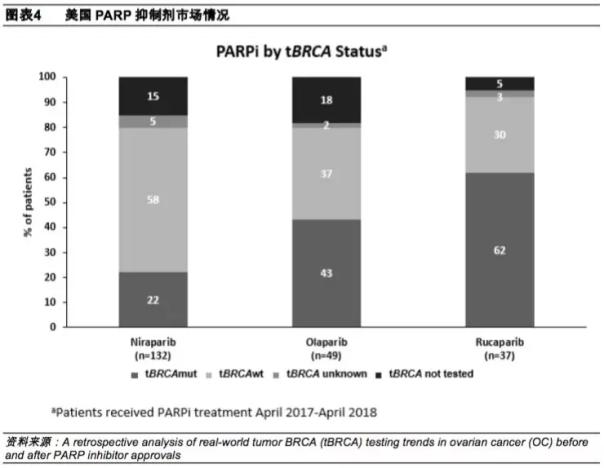
In addition, an article published on September 15 this year called A retrospective analysis of real-world tumor BRCA (tBRCA) testing trends in ovarian cancer (OC) before and after PARP inhibitor approvals shows that in the United States, with Orapa Compared with Lucapali, Nilapali is used more frequently. Since the listing, the market share has rapidly expanded to 62%, and the market potential of Olapali is huge.
Nilapali is so easy to use, and obviously it will not let it stay in ovarian cancer. In the Chinese mainland, relevant clinical trials are underway, and Ding Ding continues to increase its investment in anti-cancer drugs aimed at making it a variety of solid tumors . Let us wait and see!
Tetanus Toxoid Vaccine,Toxoid Vaccine,Hep B Immune Globulin,Immunoglobulin Injections
FOSHAN PHARMA CO., LTD. , https://www.fospharma.com